35 Prescription Drug Label Example
Editable Prescription Label Template. Fill out, securely sign, print or email your fill in the blanks prescription labels form instantly with SignNow. The most secure digital platform to get legally binding, electronically signed documents in just a few seconds. Available for PC, iOS and Android. Start a free trial now to save yourself time and money! For example, maintain a sufficient inventory of supplies necessary to support timely provision of prescription drug container labels in accessible label formats. Provide prescription medication with an accessible prescription drug label within the time frame the same prescription would be provided to patients without visual impairments.
Top 50 Common Warning Labels and Their Meanings. The medication must be swallowed whole. Because certain drugs are designed to be either fast-acting or slow-releasing, damaging the outer coating may lead to harmful damages to the body. The medication is intended for external use only. Ingesting it may lead to undesirable effects or even.
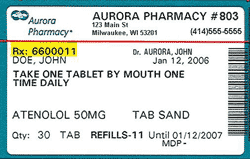
Prescription drug label example
CDER Prescription Drug Labeling Conference 2017 CDER SBIA REdI Silver Spring, MD - November 1 & 2, 2017.. DFS Section Examples • For injection: 50 mg, 100 mg, or 200 mg of drugozide as a ... The label on your prescription medication tells you how to correctly take the medicine your healthcare provider has recommended for your treatment plan. It’s very important to understand the information on this label. By taking your medication correctly, you will have the best treatment results. Appointments 216.444.2538. (prescription) drugs. Unlike over-the-counter medications, which are determined by the U.S. Food and Drug Administration (FDA) to be safe and effective for use by the general public without a doctor's prescription, legend (prescription) drugs are to be used under the supervi-sion of a licensed practitioner.
Prescription drug label example. HOW TO READ MEDICATION ORDERS DRUG LABELS PG. 6 WARNING THIS PRODUCT CONTAINS ACETAMINPHEN. AN OVERDOSE CAN DO DAMAGE TO YOUR LIVER OR CAUSE DEATH. TALK TO YOUR DOCTOR BEFORE USING ANY OVER-THE-COUNTER DRUGS. Sample PRN prescription label: Hometown Pharmacy 1234 Main Street Anywhere USA 11111 (800) 888-8888 NO 0060012-134 Best Doctor MD Best Client The label on your prescription medication tells you how to correctly take the medicine your healthcare provider has recommended for your treatment plan. It’s very important to understand the information on this label. By taking your medication correctly, you will have the best treatment results. Appointments 216.444.2538. How Not to Label Prescription Medication. Let's pretend you have a dog. Actually, you might have one. But, if you don't, pretend you went to visit a veterinarian and got some medication for your dog. According to studies, in outpatient setting, half of the adults misunderstand at least some of the prescription bottle's labels. A good way to prevent this is drafting the labels with the help of a prescription bottle label template. You can easily find the template online.
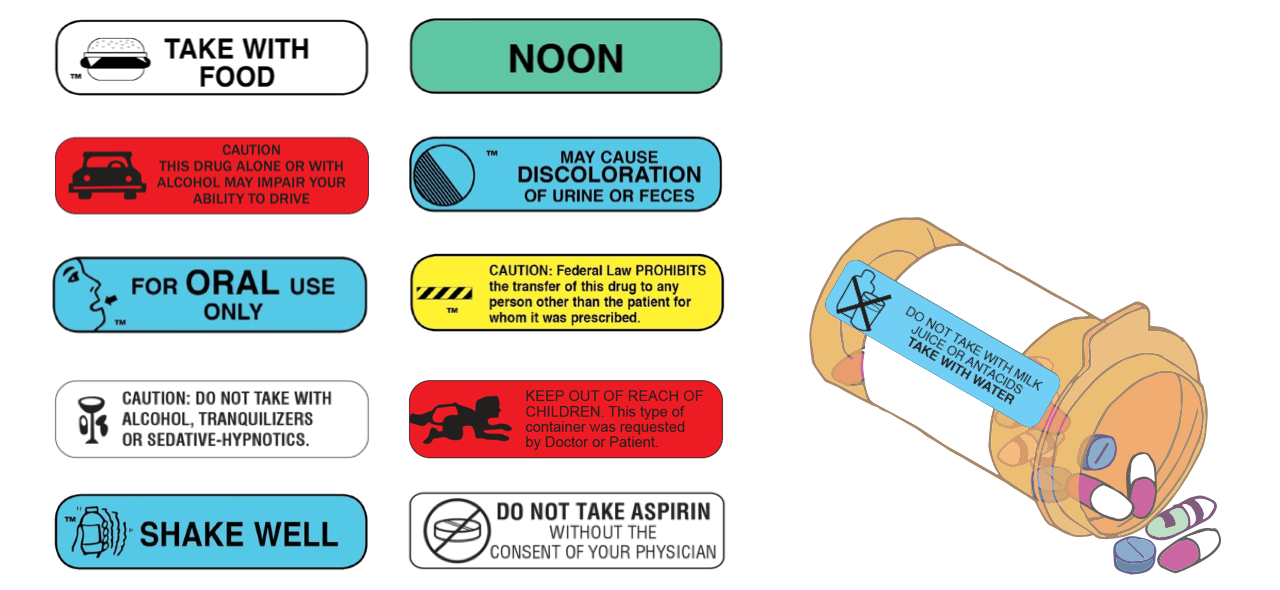
Unlike prescription labels, which cover most of the prescription vial, auxiliary labels are typically small, colorful labels that provide crucial warnings and instructions to patients using as few words as possible. Common examples of auxiliary label warnings and instructions include: "May cause drowsiness" "Keep in Refrigerator" Prescription Product Labeling. Proper labeling is one of the most important aspects of dispensing a prescription. The label must comply with state and federal regulations and should correctly and clearly convey all necessary information regarding dosage, mode of administration, and proper storage of the product. CDER Prescription Drug Labeling Conference 2017 CDER SBIA REdI Silver Spring, MD - November 1 & 2, 2017.. DFS Section Examples • For injection: 50 mg, 100 mg, or 200 mg of drugozide as a ... For example, the pharmacological classification of a drug product as an anti-emetic would be insufficient on a consumer-available non-prescription drug label; therefore, an additional indication such as "motion sickness" would be necessary.
Drug labelling is also referred to as prescription labelling, is a written, printed or graphic matter upon any drugs or any of its container, or accompanying such a drug.Drug labels seek to identify drug contents and to state specific instructions or warnings for administration, storage and disposal. 16. 1. 21 CFR 201.57(c)(9). See also the final rule "Content and Format of Labeling for Human Prescription Drug and Biological Products; Requirements for Pregnancy and Lactation Labeling" (79. (3) The drug sample shows evidence of having been stored or shipped under conditions that might adversely affect its stability, integrity, or effectiveness; (4) The drug sample is for a prescription drug product that has been recalled or is no longer marketed; or (5) The drug sample is otherwise possibly contaminated, deteriorated, or adulterated. The drug label printed on prescription drug packaging doesn't tell you all the safety information for the drug, so pharmacies will give you a pharmacy information sheet. On this sheet, look for warnings or cautions, drug uses, how to use the drug, side effects, precautions, drug interactions, overdose information and how to store the drug.
When prescription drug labeling must summarize or otherwise rely on a recommendation by an authoritative scientific body, or on a standardized methodology, scale, or technique, because the information is important to prescribing decisions, the labeling may include a reference to the source of the information.
Prescription Label Information, Translations, and Sample Labels. Translations of Pill Directions. Patient-Centered Prescription Drug Container Label Samples. Prescription Drugs: Labeling Requirements - Report to the Legislature. Statutory Requirements (4076.5) and Regulation Requirements (1707.5)
Parameters Sample Labels; Label size: 8.29 x 5.43 cm. Font family: Tahoma. Font size: 12 pt. Label size: 10.2 x 5.12 cm. Font family: Franklin Gothic Book
A brief overview of prescription drug labeling for patients and providers. SINCE 2006 , the U.S. Food and Drug Administration (FDA) has mandated both content and format of full prescribing information (FPI) and highlights for pharmaceuticals and biological products through a regulation commonly referred to as the Prescription Labeling Rule (PLR).
dosage of the drug It is essential to provide written instructions on how to take the drug All special instructions must be included, similar to the instructions on a prescription filled by a pharmacist Special Instructions can be written on a label and placed on the sample package a zip-lock bag or an inexpensive brown lunch bag.
The labels of these medications include the name and quantity of each component drug. The medication order or prescription for these drugs indicates the number of tablets, capsules, or milliliters to be used for one dose, normally not the strength of each drug. Figures 5.21 and 5.22 show examples of combination drugs. Figure 5.21 Timentin ® is.
DailyMed - NIH's labeling search tool over 130,000 labeling documents for prescription drugs (including biological products, vaccines, blood products, cellular and gene therapy products), over.
In this document, FDA-approved labeling for prescription drugs is referred to as "labeling" or "prescription drug labeling." B. Developing the Proposed Rule In recent years, there has been an increase in the length, detail, and complexity of prescription drug labeling, making it harder for health care practitioners to find specific.
Prescription Drug Label Example. Health (4 days ago) Prescription Drug Label Example.Health (4 days ago) Prescription Drug Label Example.Drugs (4 days ago) Examples Of Prescription Drug Labels.Drugs (9 days ago) Labeling guidelines for sample prescription drugs Mass.gov. Drugs (3 days ago) For example, sample medications may be placed in a larger container such as an envelope with the required.
For example, sample medications may be placed in a larger container such as an envelope with the required information written or typed on the front. Alternatively, the label may be a piece of paper affixed to the sample packaging or to a container holding the samples. The label may also be inserted inside a container holding the drug samples.
All drugs have an expiration date on them, usually prefaced by the abbreviation EXP.; you should always check that the current date is before the drug's expiration date before you give a drug to a patient. Sample Medication Labels. To better understand the different pieces of information on a drug label, let's look at a few examples: Example:
Drugs provides accurate and independent information on more than 24,000 prescription drugs, over-the-counter medicines and natural products. This material is provided for educational purposes only and is not intended for medical advice, diagnosis or treatment. Data sources include IBM Watson Micromedex (updated 11 Oct 2021), Cerner Multum™ (updated 1 Nov 2021), ASHP (updated 14 Oct 2021.
Medication Calculation Examination. Study Guide . D = Desired Dose . Q = Quantity of Solution . H = Strength on Hand . X = Unknown quantity of Drug • Sample: Physician orders 500 mg of ibuprofen (desired Dose) for a patient and you have 250 mg (Quantity on Hand) tablets (Quantity of solution) on hand.. Solution: D ÷ H x Q = X 500mg ÷ 250 mg x 1 tablet = 2 tablets
A drug sample is defined as a unit of a "prescription drug that is not intended to be sold and is intended to promote the sale of the drug."(fn3) Under the PDMA, manufacturers and distributors may distribute drug samples to practitioners licensed by law to prescribe them, or to hospital pharmacies or health care entity pharmacies at the request.
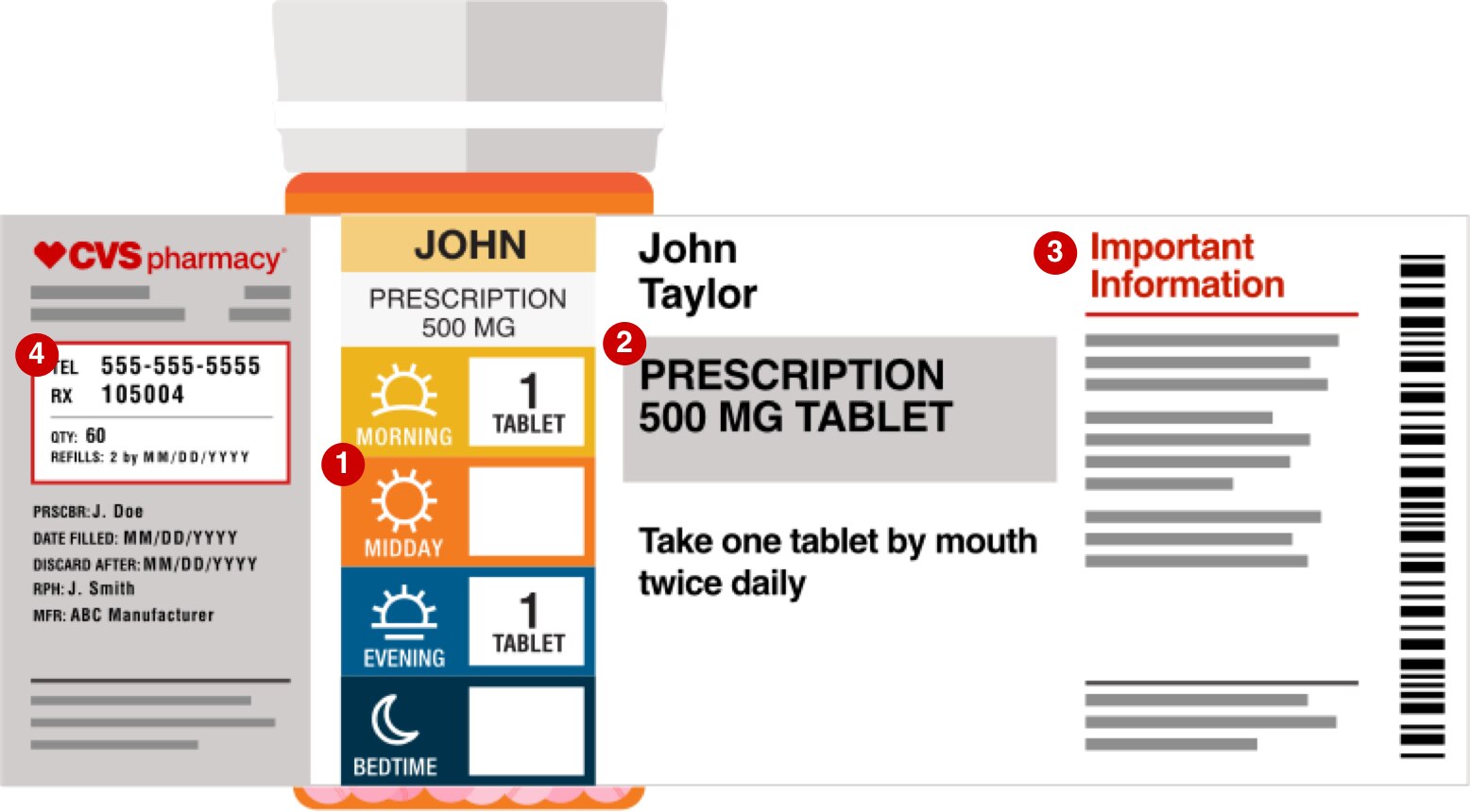
This Patient Medicine Information (PMI) includes important information such as instructions for use, and warnings about side effects and possible drug interactions. Get familiar with the different parts of your prescription drug labels with this example. Use your cursor to rollover the different sections of the label to learn more.
Sample of Prescription Drug Label. Continued . RM 1-SU: How to Read a Prescription Drug Label (Continued) Using the sample label provided, answer the following questions to determine what important information must be included on a prescription drug label. 1.
The next type of information on your prescription label is related to the drug itself. Your prescription label should have the drug…. (1) NAME. (2) STRENGTH. (3) QUANTITY. For example, the label might read: "#30 tab Lisinopril 10mg" which would mean you are supposed to have gotten 30 tablets of lisinopril 10mg.
Whether it is prescription pad design or prescription letterhead, the medical prescription formats area available in PDF. Also, online prescription label templates could be downloaded. Many examples of medical prescriptions could be seen around the web and we bring to you only the professionally crafted prescription templates.
Good Label and Package Practices Guide for Prescription Drugs 9 3 Designing Labels and Packages for Safety 3.1 Introduction Part 3 of this guide presents information on current good practices in the design and layout of a health product label, the information contained on the label, and the design or choice of package.
Healthcare providers must focus on prescription label design. If the text is too hard to read, patients will easily misunderstand their instructions. The design can also influence how much information fits onto the label. Any details that influence medication adherence must be on the bottle so patients know how to follow directions.
(prescription) drugs. Unlike over-the-counter medications, which are determined by the U.S. Food and Drug Administration (FDA) to be safe and effective for use by the general public without a doctor's prescription, legend (prescription) drugs are to be used under the supervi-sion of a licensed practitioner.




















0 Response to "35 Prescription Drug Label Example"
Post a Comment