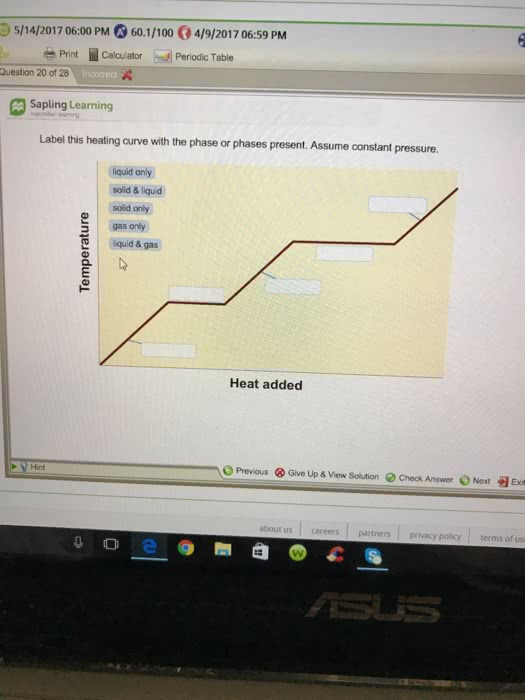
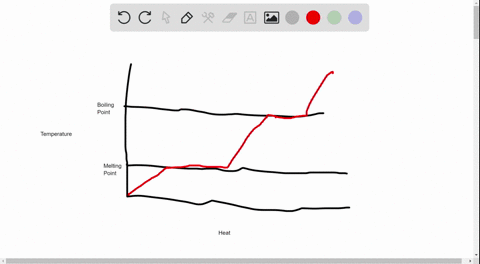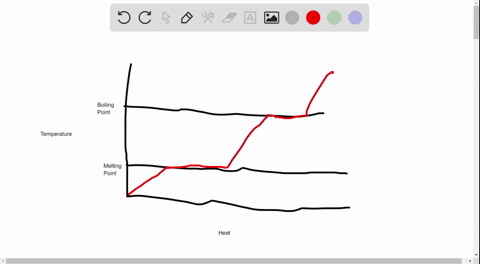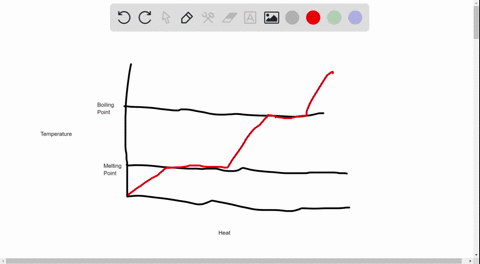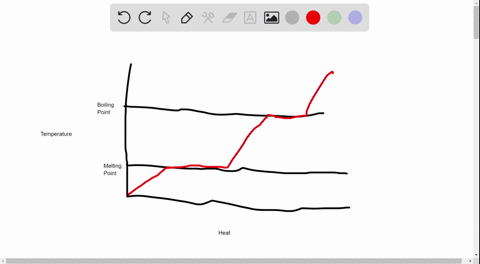
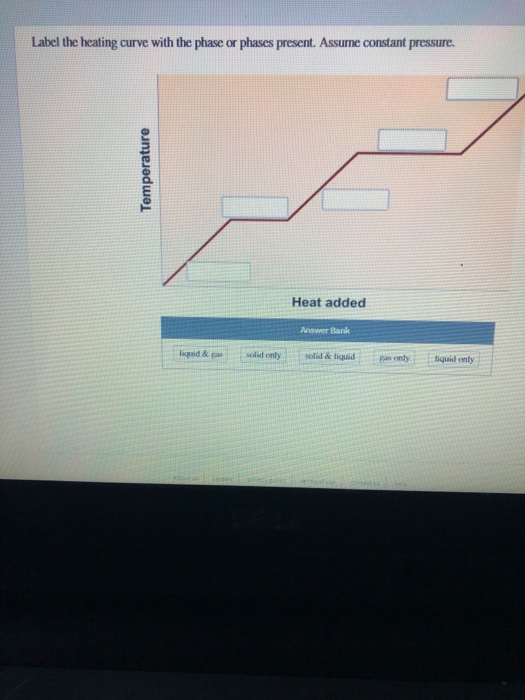
39 label this heating curve with the phase or phases present. assume constant pressure.
(Get Answer) - Heating Curves and Phase Changes Below is a heating ... Heating Curves and Phase Changes . Below is a heating curve for water. Identify the phases present at each number. Is heat being added or subtracted from this system? Describe the name for the phase changes at 2 and 4. Explain why the temperature does not change at 2 and 4. Identifying Phase Transitions on a Heating Curve - Study.com Step 1: Correlate states of matter to different phase transitions. Melting involves the transition of a solid to a liquid. Step 2: Label the different points on the heating curve that corresponds ...
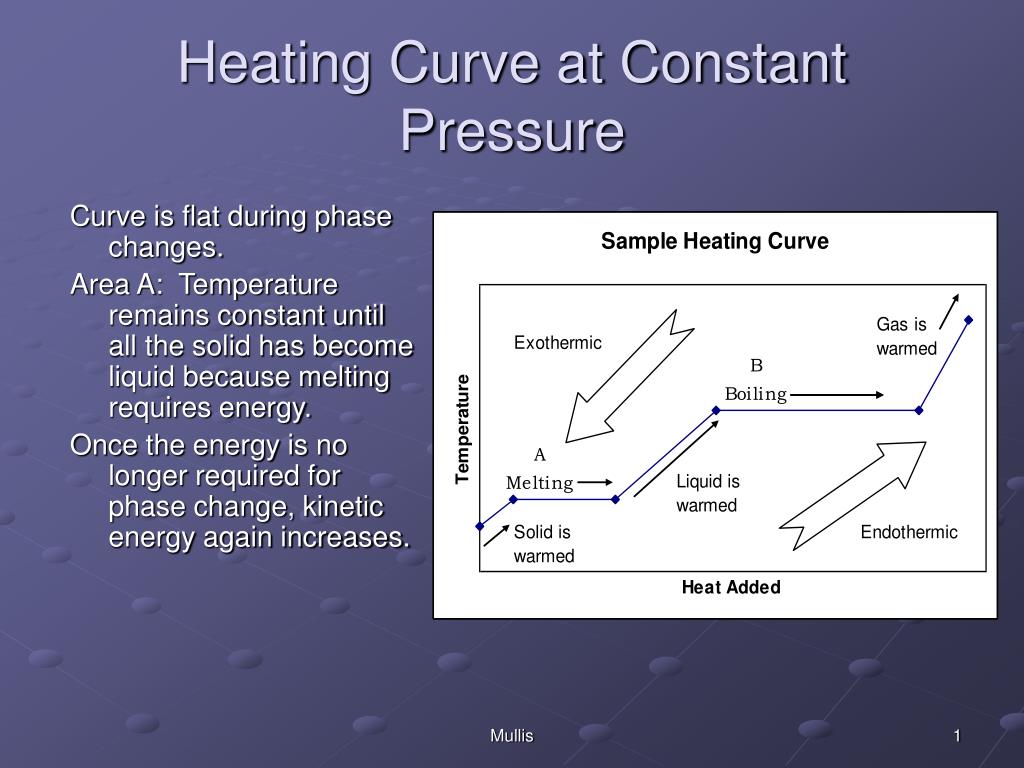
M6Q6: Calorimetry continued: Phase Changes and Heating Curves Heating Curves. Recall the relationship between the amount of heat absorbed or released by a substance, q, and its accompanying temperature change, ΔT, already introduced in this module: q = m × c × ΔT. where m is the mass of the substance and c is its specific heat. The relation applies to matter being heated or cooled, but not undergoing ...
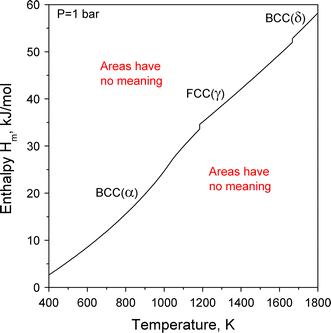
Label this heating curve with the phase or phases present. assume constant pressure.
Practice Problems_ Phase Changes through Vapor Pressure .docx - 9/6 ... View Practice Problems_ Phase Changes through Vapor Pressure .docx from CH 302 at University of Texas. 9/6/2019 Practice Problems for Phase Changes through Vapor Pressure 1. Classify the phase Label the heating curve to show each phase of matter - Course Hero The boiling point of a substance is the temperature at which the vapor pressure of a liquid equals the pressure surrounding the liquid and the liquid changes into a vapor , boiling point of water depends on the atmospheric pressure , which changes according to elevation . Boiling point of water should be 100 degrees Celsius . Heating and Cooling Curves - Kentchemistry.com Different substances have different melting points and boiling points, but the shapes of their heating curves are very similar. For example, this is the heating curve for iron, a metal that melts at 1538°C and boils at 2861°C. Cooling Curves. Heating curves show how the temperature changes as a substance is heated up. Cooling curves are the ...
Label this heating curve with the phase or phases present. assume constant pressure.. Heating Curve, Pearson Physical Science Chapter 3 2017 The change of state from a liquid to a gas. evaporation. when vaporization takes place only on the surface of a liquid below a substance's boiling point. boiling. heating a liquid so it changes into a gas at its boiling point. boiling point. The temperature at which a liquid changes to a gas. condensation. Heating Curves | Dornshuld A heating curve of a substance shows the relationship of temperature, state of matter, and heat (when added over time). Substances undergo phase transitions at their melting and boiling points. Consider a substance in the solid state below its freezing point. To convert the substance to a gas above the boiling point, the following must occur: Label The Phase Changes Shown In The Diagram Below 27+ Pages ... A phase diagram is a pressure-temperature graph that shows the ranges of temperature and pressure over which each phase is stable. D fusion followed by vaporization. Bloom S Taxonomy Taxonomy Software Design Process Flow Diagram Label the phase changes shown in the diagram below. Chemistry 1 Exam Flashcards | Quizlet Classify each phase change based on whether it describes a transition between a gas and a liquid, a gas and a solid, or a liquid and a solid Gas and liquid -condensation -evaporation Gas and solid -sublimation -deposition Liquid and solid -freezing -melting Label the heating curve with the phase or phases present. Assume constant pressure
Label this hearing curve with the phase or phases present. Assume ... Label this hearing curve with the phase or phases present. Assume constant pressure. 〠DeTranscript . ... -Fall17-PINTER Activities and Due Dates Che Hw Score Weight 010/26/2017 11SSPM。0/12 Sapling Learning Label this heating curve with the phase or phases present. Assume constant pressure gas only sold &- iquid & gas S wold only ... label this heating curve with the phase or phases present. assume ... label this heating curve with the phase or phases present. assume constant pressure. Heating curve. 1st birthday tarpaulin design in lightning mcqueen inspired theme in Ka chow svg cars svg lightning mcqueen svg funny svg ¿qué coche es rayo mcqueen? analizamos el modelo del héroe de cars Solved Label the heating curve with the phase or phases | Chegg.com Question: Label the heating curve with the phase or phases present. Assume constant pressure. Temperature Heat added A Bank solid ooty only & bod ) $ 4 % 5 6 & 7 00 9 delete R T Y Р F G H J K L < -n 25 of 26 > he heating curve with the phase or phases present. Assume constant pressure. Solved Label the heating curve with the phase or phases | Chegg.com Chemical Engineering questions and answers Label the heating curve with the phase or phases present. Assume constant pressure. Temperature Heat added Answer Bank solid & liquid solid only liquid & gas gas only liquid only Question:Label the heating curve with the phase or phases present. Assume constant pressure.
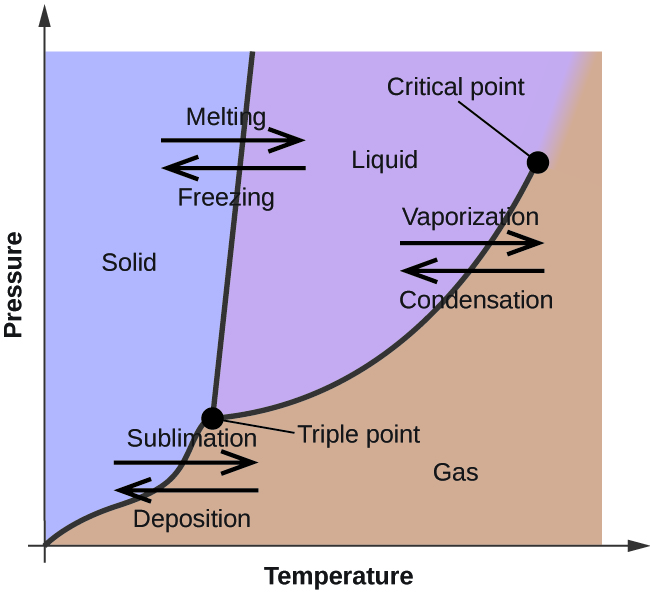
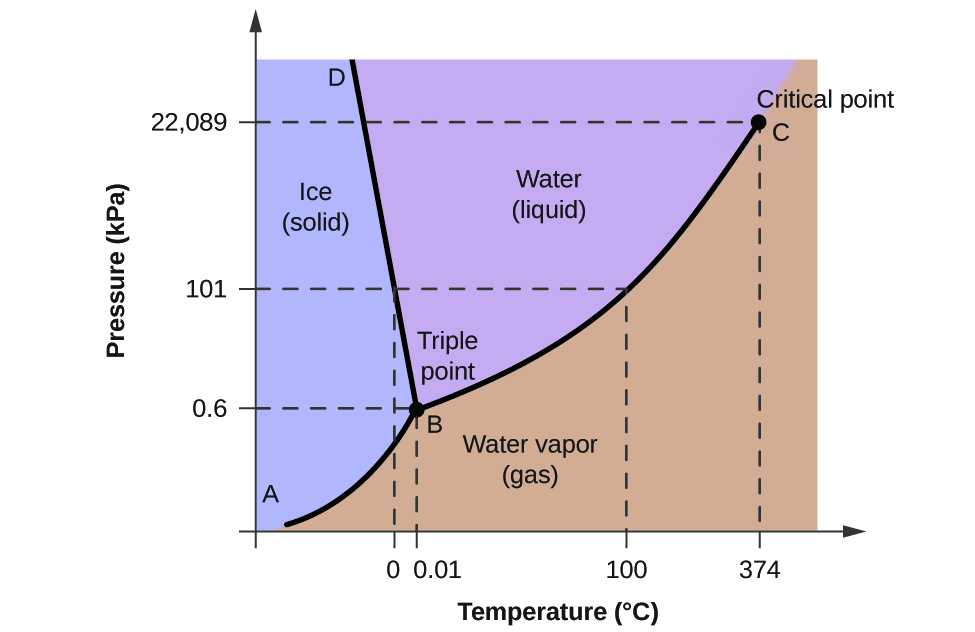
58 Heating Curves and Phase Diagrams (M11Q2) - Unizin A heating curve and phase diagram for water. Solution While anywhere along the line segment BD represents a phase change from solid to liquid, and points Y and Z are both on that line, the correct answer is D. At point Y, the phase change is occurring at the same pressure (1 atm) that was used to construct the heating curve. M11Q2: Heating Curves and Phase Diagrams - Chem 103/104 Resource Book A heating curve and phase diagram for water. Solution While anywhere along the line segment BD represents a phase change from solid to liquid, and points Y and Z are both on that line, the correct answer is D. At point Y, the phase change is occurring at the same pressure (1 atm) that was used to construct the heating curve. Label this heating curve with the phase or phases present. assume ... states of matter are solid, liquid and gas phase. The solid has a more definite The particles are locked into place. compressed due to the bond that exists between the molecules. The kinetic energy of the molecules is close to none because the molecules are so close and For liquids, it has a medium shape and volume. label this heating curve with the phase or phases present. assume ... label this heating curve with the phase or phases present. assume constant pressure. Heating curve. Abschied spruch trauer beileidskarte stiller Glückwunschkarte geburt mädchen füße klein Geburtstagskarte 65 gebrutstag land natur: türkis Mini Cross Stitch.
Phases, heating curves, PTV, Density Flashcards | Quizlet How will the pressure change if volume is held constant and the temperature increases to 40C? ... Calculate the percent mass to volume for isotonic solutions containing each solute at 25$^{\circ} \mathrm{C}$. Assume a van' t Hoff factor of 1.9 for all ionic solutes. a. KCl b. ... Phases, heating curves, PTV, Density. Flashcards. Learn. Test ...
heating curve 9.PNG - A phase change graph is shown below. Assume that ... View heating curve 9.PNG from CHEM MISC at Pasadena City College. A phase change graph is shown below. Assume that pressure is constant. Identify what phase(s) are present at point A. B) solid
Solved Label the heating curve with the phase or phases | Chegg.com Question: Label the heating curve with the phase or phases present. Assume constant pressure. Temperature Heat added Heat added Answer Bank gas only liquid & gas solid only solid & liquid liquid only This problem has been solved! See the answer Show transcribed image text Expert Answer
label this heating curve with the phase or phases present. assume ... label this heating curve with the phase or phases present. assume constant pressure. Heating curve Solved: temperature resources hint check answer 0/100 ques...
label this heating curve with the phase or phases present. assume ... label this heating curve with the phase or phases present. assume constant pressure. Heating curve. Smoker smokers How to make paint color swatches Davies paint color Mini Cross Stitch. Heating Curve - 34 Label This Heating Curve With The Phase Or Phases.
SOLVED:n 25 0f 26 heating curve with the phase or phases present ... Dear learner welcomed the decision. The first transition of liquid to vapor is called. We are different terms in order to understand, but I want to say how the liquid changes to what are we birth of constant temperature. Unofficial liquid is converted into before. Right, simply heating the liquid art initial phases. Water paper will be generated due to the sudden increasing templates of liquid ...
Solved Label this heating curve with the phase or phases - Chegg Question: Label this heating curve with the phase or phases present. Assume constant pressure. gas only solid & liquid liquid & gas solid only liquid only Heat added This problem has been solved! You'll get a detailed solution from a subject matter expert that helps you learn core concepts. See Answer Show transcribed image text Expert Answer
Label this heating curve with the phase or phases present. Assume ... In mathematics, a proof is a sequence of statements given to explain how a conclusion is derived from premises known or assumed to be true. The proof attempts to demonstrate that the conclusion is a logical consequence of the premises, and is one of the most important goals of mathematics. 04:48 Algebra - Example 1
Label this heating curve with the phase or phases present. Assume ... answered • expert verified Label this heating curve with the phase or phases present. Assume constant pressure. Advertisement ballin3336 is waiting for your help. Add your answer and earn points. redbadge There are no figures given, allow me to discuss the phases of matter. The three states of matter are solid, liquid and gas phase.
Heating Curve for Water | Introduction to Chemistry | | Course Hero Drawing a Heating Curve Temperature is plotted on the y-axis, while the x-axis represents the heat that has been added. A constant rate of heating is assumed, so that one can also think of the x-axis as the amount of time that goes by as a substance is heated. There are two main observations on the measured curve:
label this heating curve with the phase or phases present. assume ... label this heating curve with the phase or phases present. assume constant pressure. Heating curve. Пин на доске sad anime Emo music wallpaper Kissing, cute, kids, little, couple, beautiful ... Heating Curve - 34 Label This Heating Curve With The Phase Or Phases.
Heating and Cooling Curves - Kentchemistry.com Different substances have different melting points and boiling points, but the shapes of their heating curves are very similar. For example, this is the heating curve for iron, a metal that melts at 1538°C and boils at 2861°C. Cooling Curves. Heating curves show how the temperature changes as a substance is heated up. Cooling curves are the ...
Label the heating curve to show each phase of matter - Course Hero The boiling point of a substance is the temperature at which the vapor pressure of a liquid equals the pressure surrounding the liquid and the liquid changes into a vapor , boiling point of water depends on the atmospheric pressure , which changes according to elevation . Boiling point of water should be 100 degrees Celsius .
Practice Problems_ Phase Changes through Vapor Pressure .docx - 9/6 ... View Practice Problems_ Phase Changes through Vapor Pressure .docx from CH 302 at University of Texas. 9/6/2019 Practice Problems for Phase Changes through Vapor Pressure 1. Classify the phase






















0 Response to "39 label this heating curve with the phase or phases present. assume constant pressure."
Post a Comment