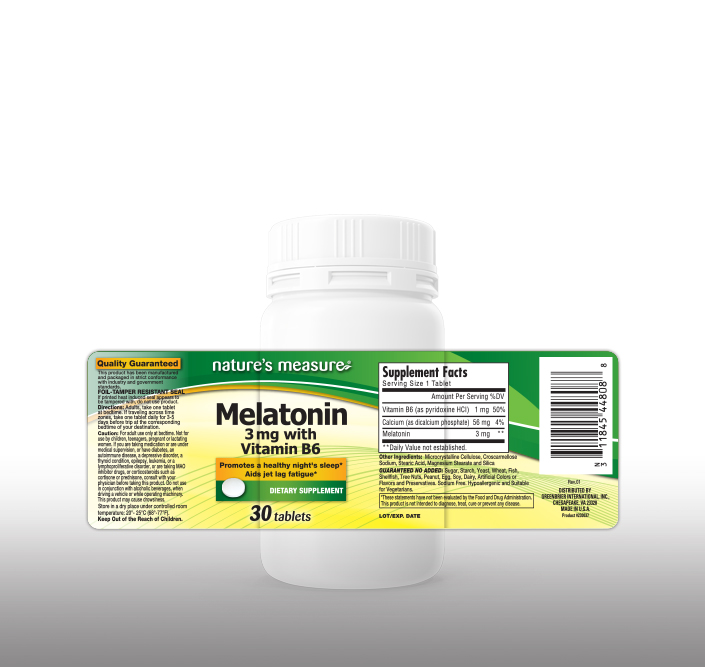
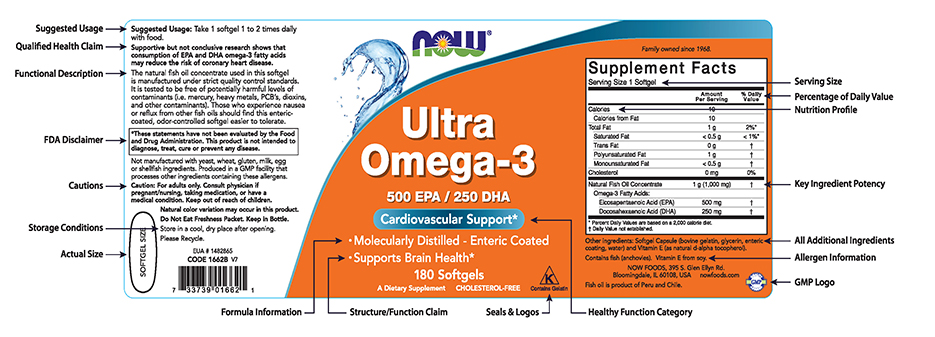
42 dietary supplement label requirements
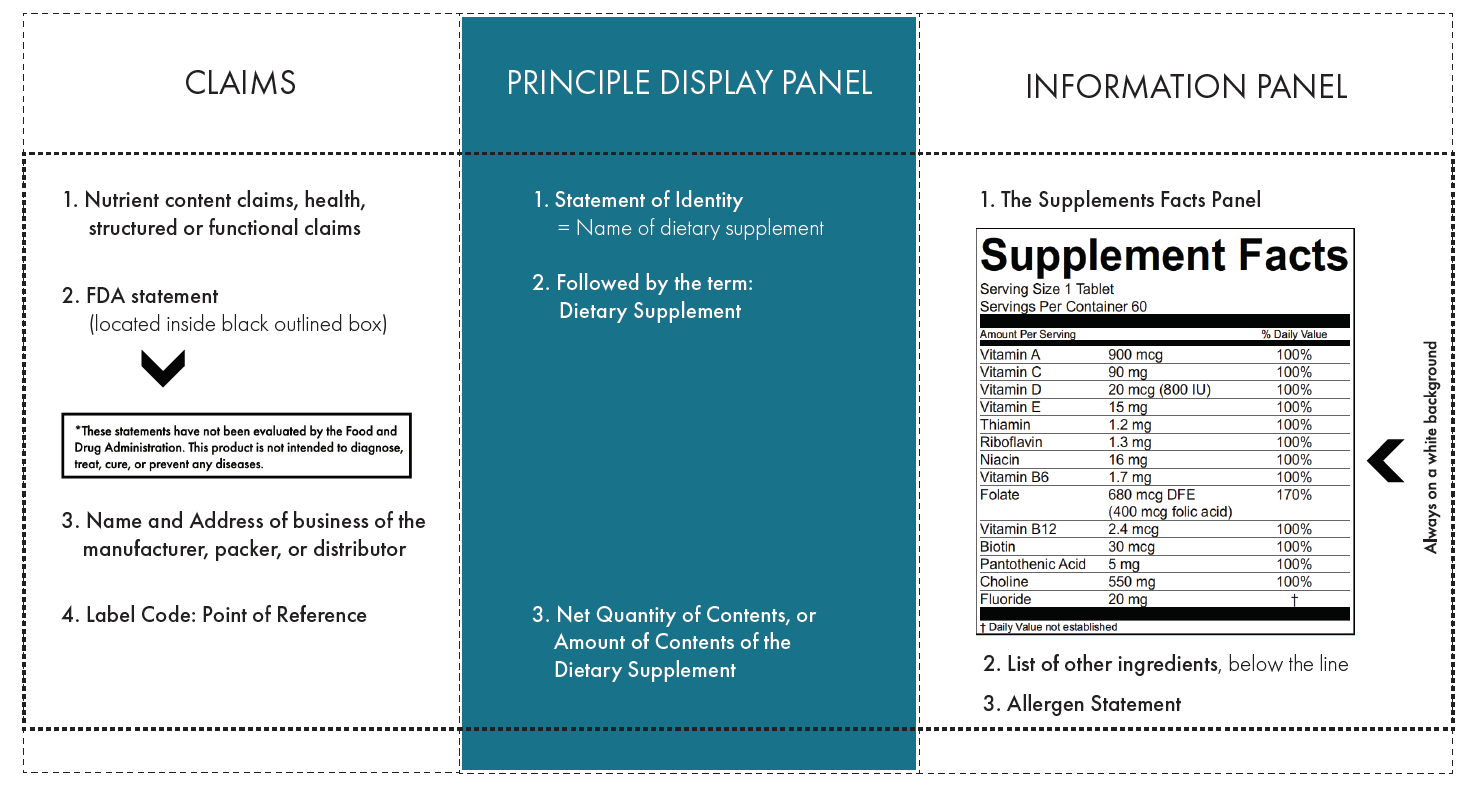
FDA Dietary Supplement Labeling Requirements: Comply or Die May 1, 2018 ... FDA Dietary Supplement Labeling Requirements: Comply or Die · the statement of identity (name of the dietary supplement); · the net quantity of ... Labeling Dietary Supplements The U.S. Food and Drug Administration regulations require that dietary supplement labeling include a descriptive name of the product stating that it is a ...
How to Create an FDA Approved Supplement Facts Label - Sttark Jul 13, 2020 ... FDA Supplement Labeling Regulations · Statement of Identity — This is the official name of the supplement. · Net Quantity of Contents Statement ...

Dietary supplement label requirements
Food supplements – EU labelling rules - Your Europe Labelling requirements · portion of the product recommended for daily consumption · warning not to exceed the recommended daily dose · statement that food ... Dietary Supplement Labeling - American Bar Association Apr 10, 2018 ... The label of a dietary supplement product is required to be truthful and not misleading. If the label does not meet this requirement, the FDA ... Dietary Supplement Labeling Guide - FDA Apr 1, 2005 ... The Dietary Supplement Health and Education Act of 1994 (the DSHEA) amended the act, in part, by defining "dietary supplements," adding specific ...
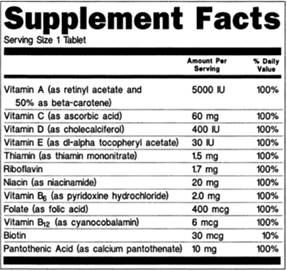
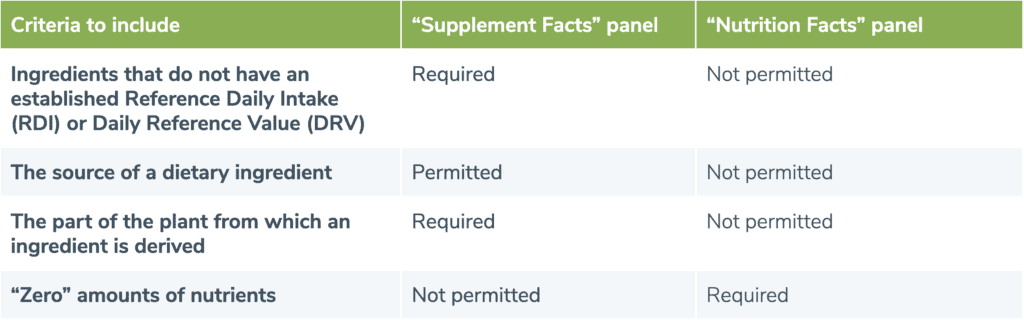
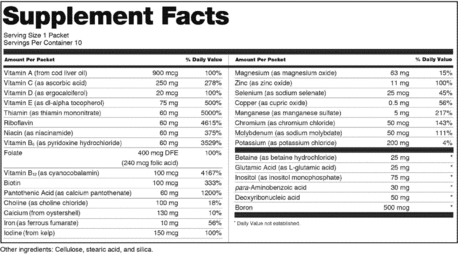
Dietary supplement label requirements. Dietary Supplement Labeling: Everything You Need to Know Apr 2, 2022 ... You are required to include the name and contact information of the supplement manufacturer, packer or distributor. If there could be hazards ... 21 CFR § 101.36 - Nutrition labeling of dietary supplements. (1) The title, “Supplement Facts,” shall be set in a type size larger than all other print size in the nutrition label and, unless impractical, shall be set ... Dietary Supplement Labeling Guide: Chapter I. General ... - FDA Apr 1, 2005 ... Five statements are required: 1) the statement of identity (name of the dietary supplement), 2) the net quantity of contents statement (amount ... Dietary Supplement Labels: Key Elements (OEI-01-01-00120 Given the limited requirements governing dietary supplement labels, FDA officials have expressed an interest in our presenting a vision for the kind of ...
Dietary Supplement Labeling Guide - FDA Apr 1, 2005 ... The Dietary Supplement Health and Education Act of 1994 (the DSHEA) amended the act, in part, by defining "dietary supplements," adding specific ... Dietary Supplement Labeling - American Bar Association Apr 10, 2018 ... The label of a dietary supplement product is required to be truthful and not misleading. If the label does not meet this requirement, the FDA ... Food supplements – EU labelling rules - Your Europe Labelling requirements · portion of the product recommended for daily consumption · warning not to exceed the recommended daily dose · statement that food ...







![Dietary Supplement Labeling Guide for New Brand Owners [PDF]](https://cdn2.hubspot.net/hubfs/2062774/Imported_Blog_Media/Six-Supplement-Label-Design-Tips-For-New-Brand-Owners.png)

























0 Response to "42 dietary supplement label requirements"
Post a Comment