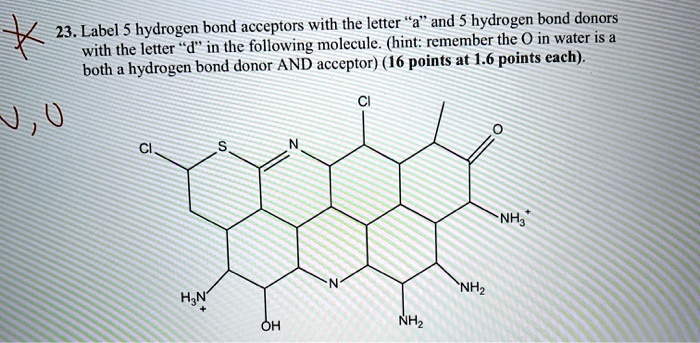
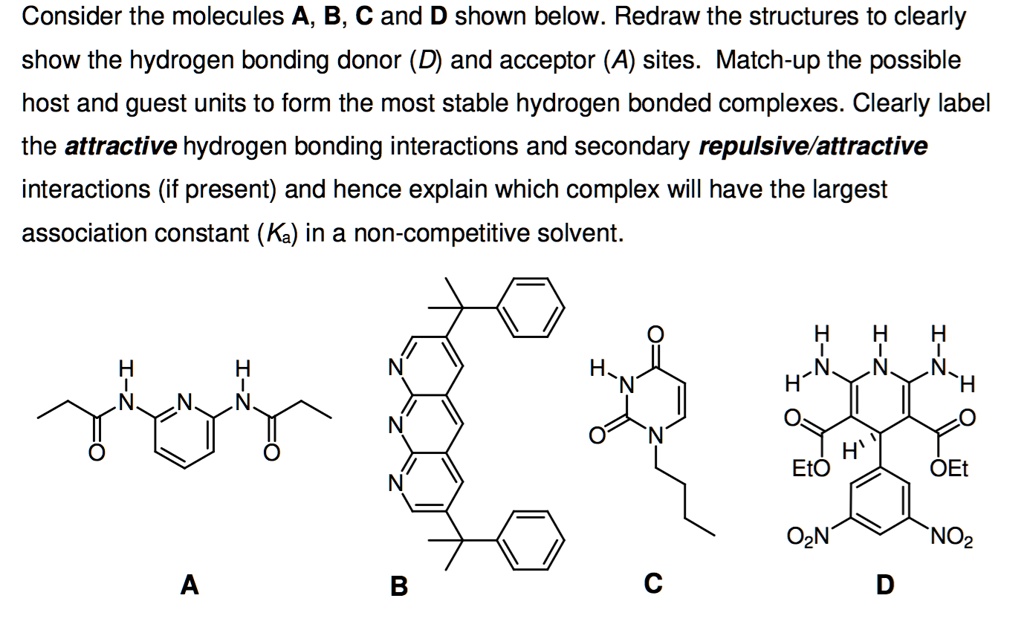
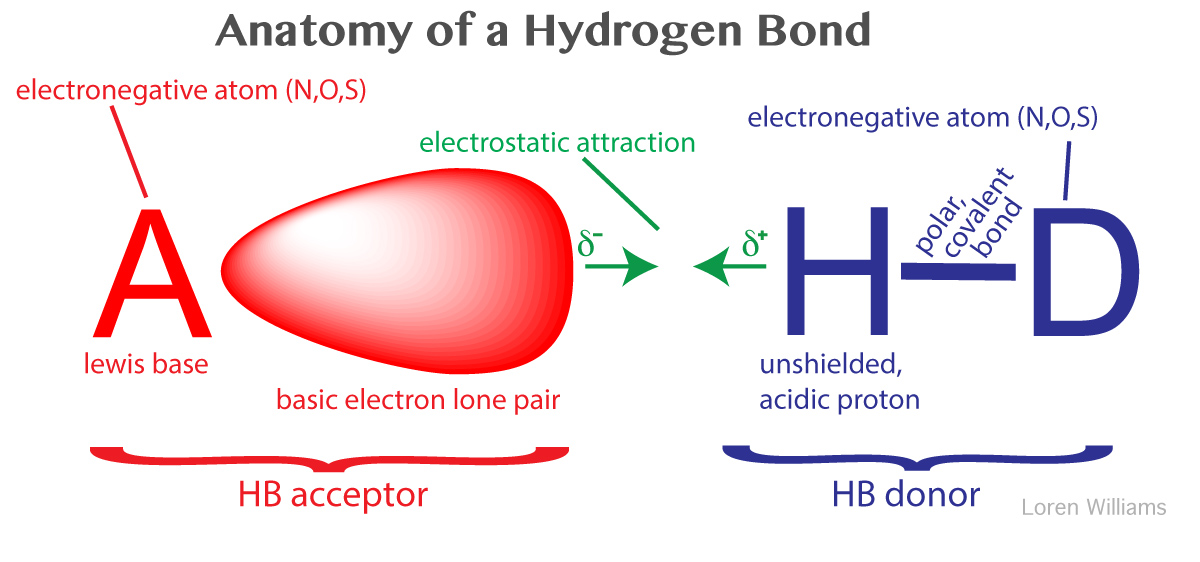
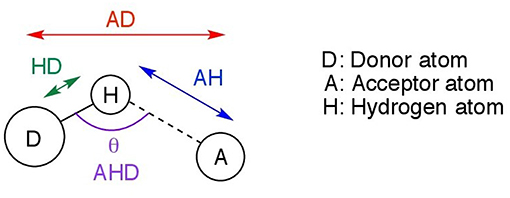
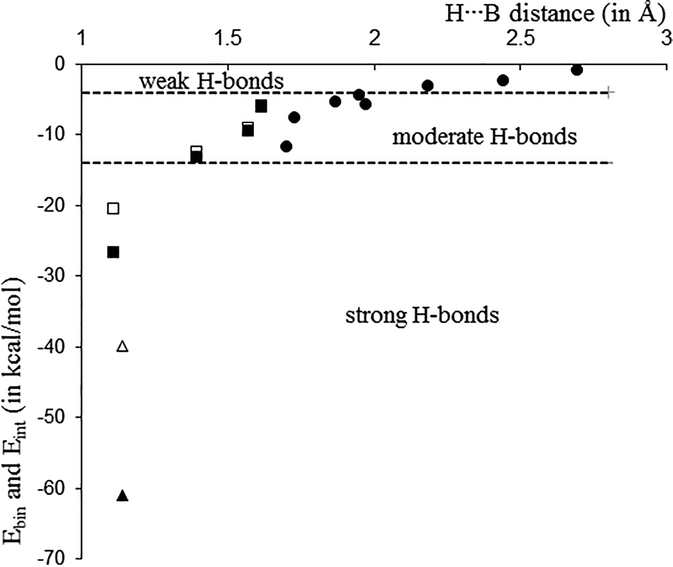
43 clearly label the hydrogen-bond donor and acceptor atoms.
pubs.acs.org › toc › jacsatJournal of the American Chemical Society | Vol 144, No 12 Two functional heterojunctions, Pd/NC and MoC/NC, are integrated together to construct unique mass (reactants and intermediates) exchange paths with interface charge-density-dependent transfer efficiencies, successfully triggering the capture and activation of CO2 and H2 molecules for mild and cascade methylation of amines. View the article. clearly label the hydrogen-bond donor and acceptor atoms. Rapid ... clearly label the hydrogen-bond donor and acceptor atoms. Rapid parameterization of small molecules using the force field toolkit ... 35 Clearly Label The Hydrogen Bond Donor And Acceptor Atoms. - Labels. 35 Clearly Label The Hydrogen Bond Donor And Acceptor Atoms. - Labels. Random Posts. Shannon Bream Measure Bream Sheldon Salary Affair;
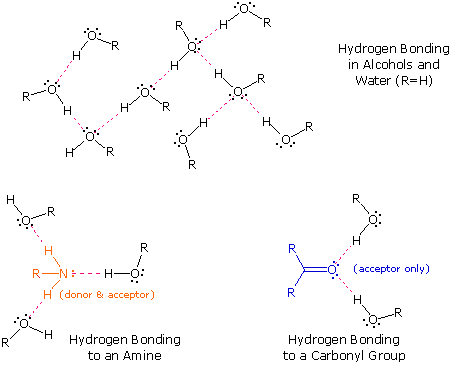
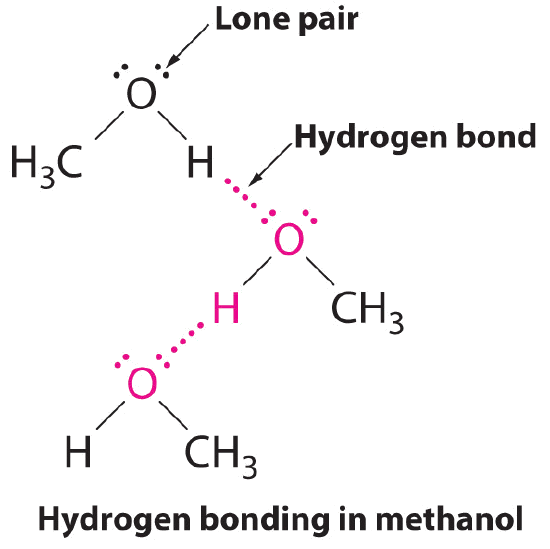
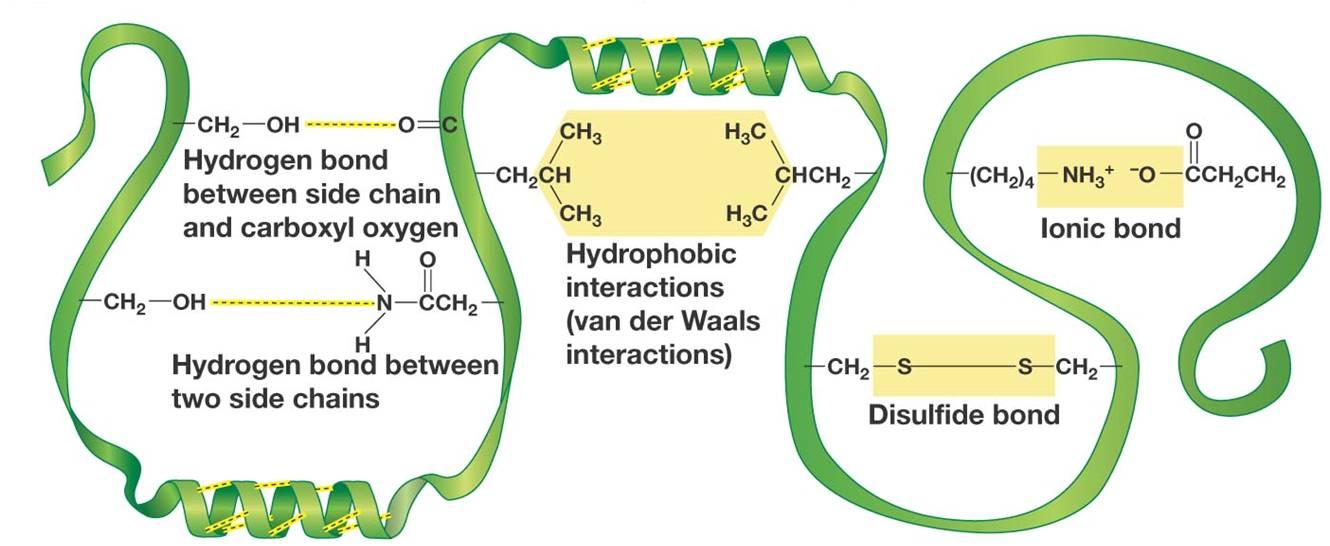
Difference Between Hydrogen Bond Donor and Acceptor A hydrogen bond forms between a hydrogen donor and acceptor. The key difference between hydrogen bond donor and acceptor is that hydrogen bond donor contains the hydrogen atom which participates in the hydrogen bond formation whereas hydrogen bond acceptor contains lone electron pairs.
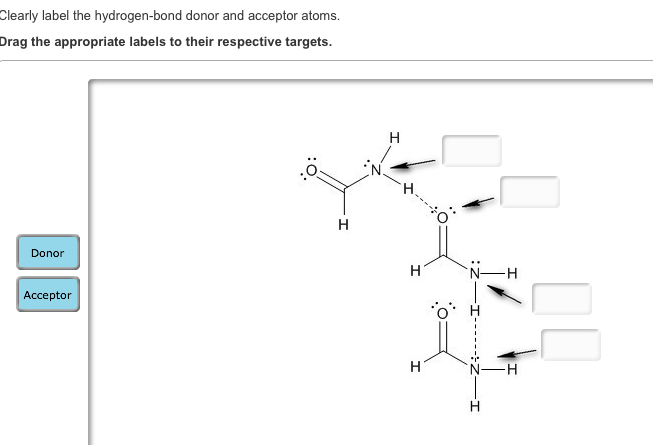
Clearly label the hydrogen-bond donor and acceptor atoms.
Mastering Biochemistry Chapter 2 Post-Lecture Hydrogen Bonding/ Acid ... A hydrogen-bond donor is the atom to which the hydrogen is covalently bonded. In formamide, the hydrogen atoms are covalently bonded to the carbon atom of the aldehyde group and the nitrogen atom of the amine group. However, only N is highly electronegative allowing the attached hydrogen to participate in hydrogen bonding. Which atoms are often hydrogen bond acceptors? - TimesMojo Hydrogen bonds occur when a "donor" atom donates its covalently bonded hydrogen atom to an electronegative "acceptor" atom. The oxygen in -OH (e.g. Ser, Thr, Tyr), HOH, and the nitrogen in -NH3+(as in Lys, Arg) or -NH- (as in the main chain peptide bond, Trp, His, Arg, nucleotide bases) are typical donors. clearly label the hydrogen-bond donor and acceptor atoms. Get the detailed answer: clearly label the hydrogen-bond donor and acceptor atoms. OneClass: clearly label the hydrogen-bond donor and acceptor atoms. 🏷️ LIMITED TIME OFFER: GET 20% OFF GRADE+ YEARLY SUBSCRIPTION →
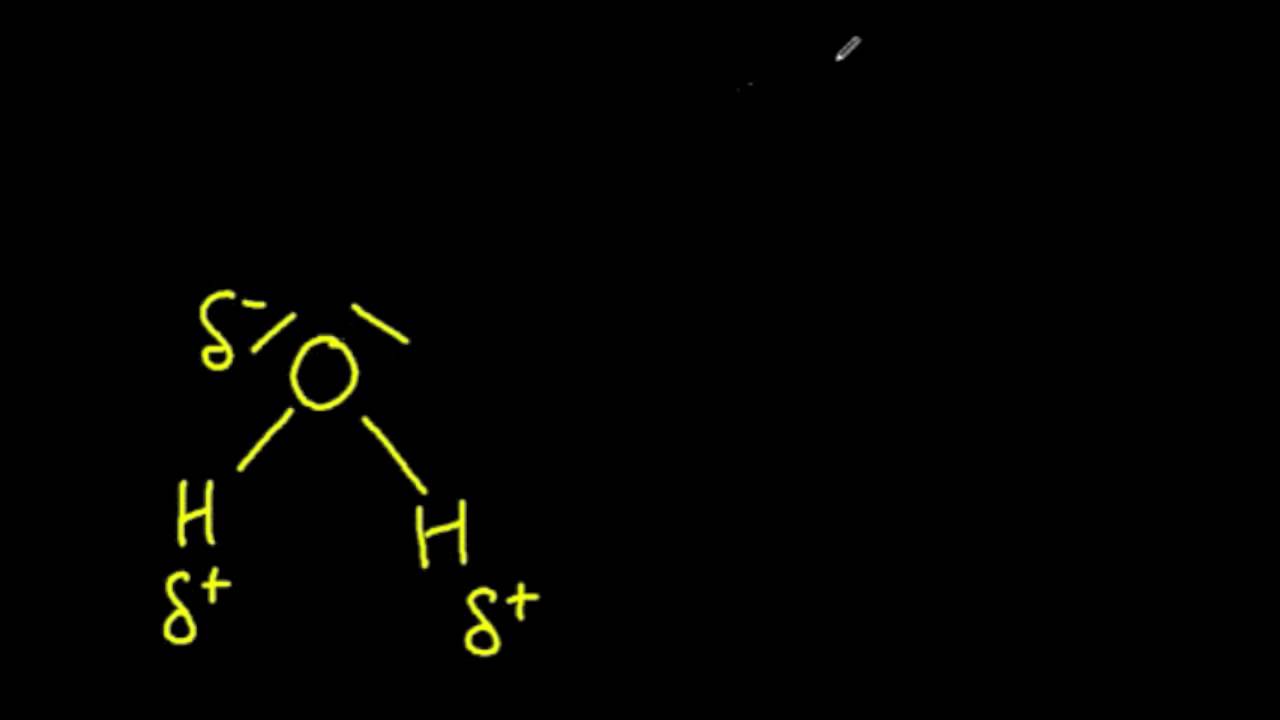
Clearly label the hydrogen-bond donor and acceptor atoms.. What groups are hydrogen bond acceptors? - Short-Fact Hydrogen bond acceptor: The atom, ion, or molecule component of a hydrogen bond which does not supply the bridging (shared) hydrogen atom. In this hydrogen bond between water and ammonia, ammonia is the hydrogen bond acceptor (shown in red), and water is the hydrogen bond donor. Which functional groups are capable of hydrogen bonding? What is the Difference Between Hydrogen Bond Donor and Acceptor The key difference between hydrogen bond donor and acceptor is that hydrogen bond donor contains the hydrogen atom which participates in the hydrogen bond formation whereas hydrogen bond acceptor contains lone electron pairs. A hydrogen bond is a weak bond between two molecules resulting from an electrostatic attraction between a proton in one ... 35 Clearly Label The Hydrogen Bond Donor And Acceptor Atoms. - Labels 35 Clearly Label The Hydrogen Bond Donor And Acceptor Atoms. - Labels is free image that you can download for free in My Awesome Site. This 35 Clearly Label The Hydrogen Bond Donor And Acceptor Atoms. - Labels has 700px x 198px resolution. Iucr donor hydrogen acceptor atoms Image type is png. Identifying Hydrogen Bond Donors & Acceptors - YouTube Practice identifying organic molecules as hydrogen bond donors and/or acceptors.
pubchem.ncbi.nlm.nih.gov › compound › paclitaxelPaclitaxel | C47H51NO14 - PubChem Paclitaxel is an antineoplastic agent which acts by inhibitor of cellular mitosis and which currently plays a central role in the therapy of ovarian, breast, and lung cancer. Therapy with paclitaxel has been associated with a low rate of serum enzyme elevations, but has not been clearly linked to cases of clinically apparent acute liver injury. Biochemistry Timed Mastering Assignments (Ch 1, 2, 3, 4) Nucleic acids (DNA/RNA) are responsible for information storage, transmission, and expression. Which of the following statements details the role of lipids? 1. some serve as a fuel source 2. some provide structure to membranes 3. some transport small molecules and ions across membranes. 4. some are precursors to steroids. 1, 2, and 4 are correct. Question : Clearly label the hydrogen-bond donor and acceptor atoms ... Question: Clearly label the hydrogen-bond donor and acceptor atoms. Drag the appropriate labels to their respective targets. This problem has been solved! You'll get a detailed solution from a subject matter expert that helps you learn core concepts. See Answer Clearly label the hydrogen-bond donor and acceptor atoms. Show transcribed image text pubchem.ncbi.nlm.nih.gov › compound › vinblastineVinblastine | C46H58N4O9 - PubChem Vinblastine sulfate is indicated in the palliative treatment of the following: Frequently Responsive Malignancies: Generalized Hodgkin's disease (Stages III and IV, Ann Arbor modification of Rye staging system), Lymphocytic lymphoma (nodular and diffuse, poorly and well differentiated), histiocytic lymphoma, mycosis fungoides (advanced stages), advanced carcinoma of the testis, Kaposi's ...
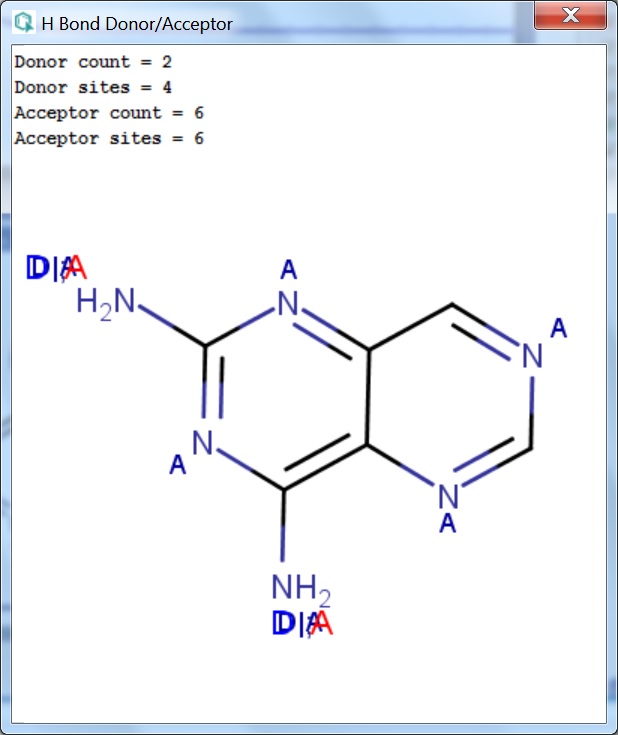
portlandpress.com › essaysbiochem › articleEssential chemistry for biochemists | Essays in Biochemistry ... Sep 26, 2017 · (ii) The ‘C’ label for carbon atoms is omitted. (iii) The ‘H’ label for hydrogen atoms is omitted only for hydrogen atoms bonded to carbon. When interpreting a skeletal formula, we assume that carbon will make four bonds; if a carbon appears to make fewer than four bonds in the skeletal representation, then the ‘missing’ bonds are ... What makes a hydrogen donor? - Studybuff Hydrogen bond donors are electronegative atoms (typically F, N, or O, but other atoms can participate) that donate hydrogen in a non-covalent way--to another. ... Donor sites = the sum of the H atoms connected to the donor atoms. Acceptor count = the sum of the acceptor atoms. An acceptor atom always has a lone electron pair/lone electron pairs ... Ch 2 Flashcards | Quizlet Clearly label the hydrogen-bond donor and acceptor atoms. ... The atom to which the hydrogen atom is covalently bonded is the hydrogen-bond donor. 3) The distance between the covalently bound H atom and its hydrogen-bonding donor is the sum of its van der Waals radii. ... Water is both a hydrogen bond donor and acceptor. True. T/F: Amphipathic ... What is a hydrogen bond acceptor and donor? Are all hydrogen bond donors acceptors? Because there is an equivalent partial negative charge on the atom bonded to hydrogen (mostly NOF), this atom can accept H-bonds from another atoms. Since H-bond donors are ALWAYS H-bond acceptors, we simplify communication to "H-bond donor". There are two H-bonding interactions for H-bond donors.
› journals › jnmBiosynthesis of Nanoparticles by Microorganisms and Their ... May 16, 2011 · The development of eco-friendly technologies in material synthesis is of considerable importance to expand their biological applications. Nowadays, a variety of inorganic nanoparticles with well-defined chemical composition, size, and morphology have been synthesized by using different microorganisms, and their applications in many cutting-edge technological areas have been explored. This ...
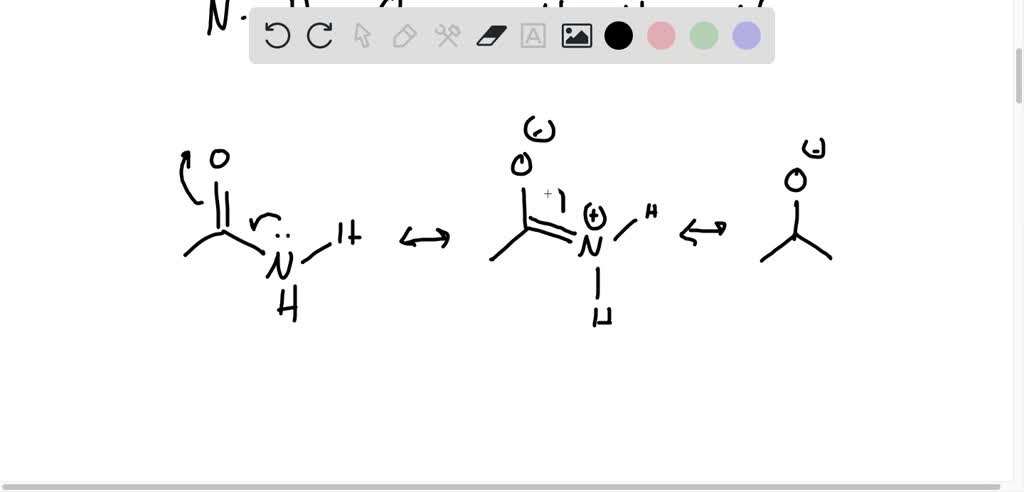
Answered: Draw two different possible… | bartleby Draw two different possible hydrogen-bonding interactions between two molecules of formamide (HCONH 2). Clearly label the hydrogen-bond donor and acceptor atoms. Which of these two possible hydrogen-bonding interactions is more likely to occur? (Hint: Consider resonance structures for formamide.)
clearly label the hydrogen-bond donor and acceptor atoms. Rapid ... Donor atoms acceptor. 34 clearly label the hydrogen bond donor and acceptor atoms.. 35 clearly label the hydrogen bond donor and acceptor atoms. ← olifant tekening kleur Volwassenen mindful boek kleurboek deltas 2891 trotsemoeders olifant tekening zwart wit Tekening van druiven vector illustratie. illustratie bestaande uit wijn →
pubs.acs.org › doi › 10Carbon Dots: A New Type of Carbon-Based Nanomaterial with ... Carbon dots (CDs), as a new type of carbon-based nanomaterial, have attracted broad research interest for years, because of their diverse physicochemical properties and favorable attributes like good biocompatibility, unique optical properties, low cost, ecofriendliness, abundant functional groups (e.g., amino, hydroxyl, carboxyl), high stability, and electron mobility. In this Outlook, we ...
Bio Chem Homework 3 Flashcards | Quizlet A hydrogen bond is equivalent to a covalent bond. *A hydrogen atom acquires a partial positive charge when it is covalently bonded to an F atom. *Hydrogen bonding occurs when a hydrogen atom is covalently bonded to an N, O, or F atom. Identify which of the following molecules can exhibit hydrogen bonding as a pure liquid. Check all that apply.
› docs › RDKit_BookThe RDKit Book — The RDKit 2022.09.1 documentation Chemical features are defined by a Feature Type and a Feature Family. The Feature Family is a general classification of the feature (such as “Hydrogen-bond Donor” or “Aromatic”) while the Feature Type provides additional, higher-resolution, information about features. Pharmacophore matching is done using Feature Family’s.
clearly label the hydrogen-bond donor and acceptor atoms. Get the detailed answer: clearly label the hydrogen-bond donor and acceptor atoms. OneClass: clearly label the hydrogen-bond donor and acceptor atoms. 🏷️ LIMITED TIME OFFER: GET 20% OFF GRADE+ YEARLY SUBSCRIPTION →
Which atoms are often hydrogen bond acceptors? - TimesMojo Hydrogen bonds occur when a "donor" atom donates its covalently bonded hydrogen atom to an electronegative "acceptor" atom. The oxygen in -OH (e.g. Ser, Thr, Tyr), HOH, and the nitrogen in -NH3+(as in Lys, Arg) or -NH- (as in the main chain peptide bond, Trp, His, Arg, nucleotide bases) are typical donors.
Mastering Biochemistry Chapter 2 Post-Lecture Hydrogen Bonding/ Acid ... A hydrogen-bond donor is the atom to which the hydrogen is covalently bonded. In formamide, the hydrogen atoms are covalently bonded to the carbon atom of the aldehyde group and the nitrogen atom of the amine group. However, only N is highly electronegative allowing the attached hydrogen to participate in hydrogen bonding.









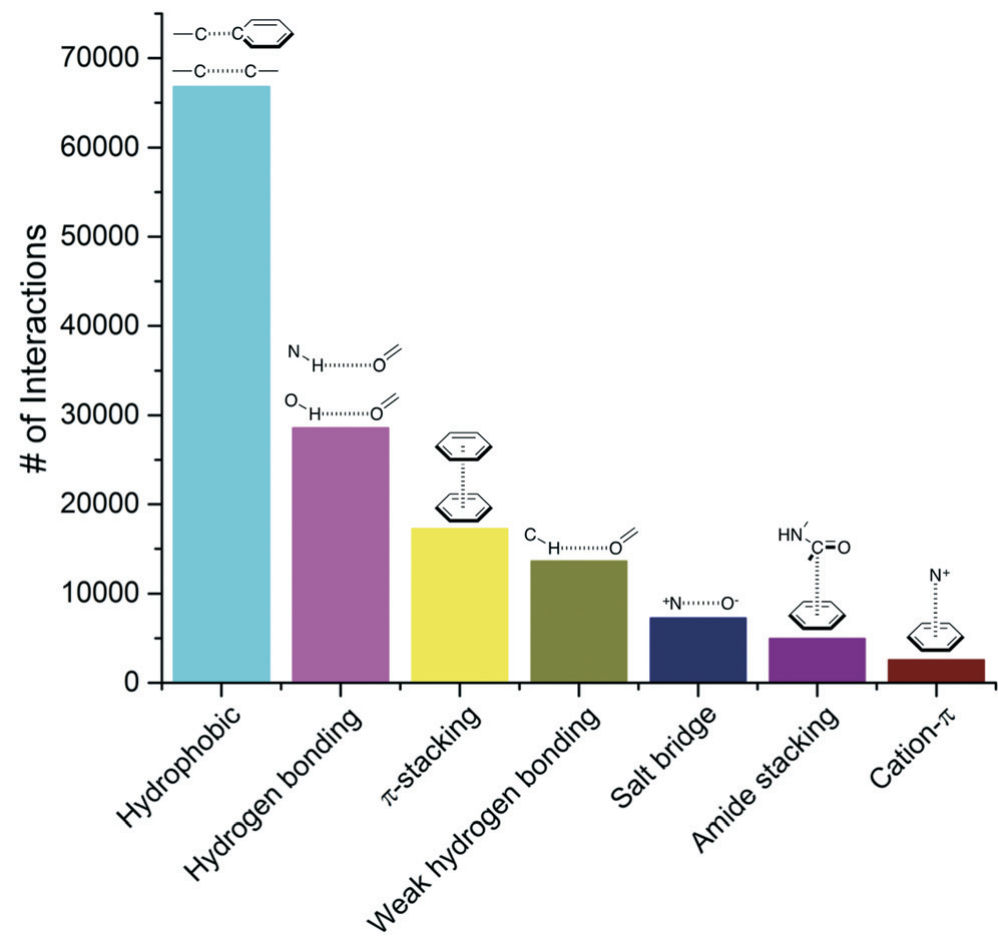




0 Response to "43 clearly label the hydrogen-bond donor and acceptor atoms."
Post a Comment