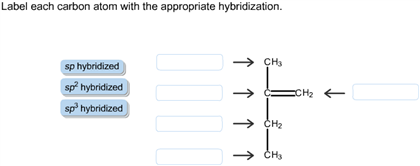
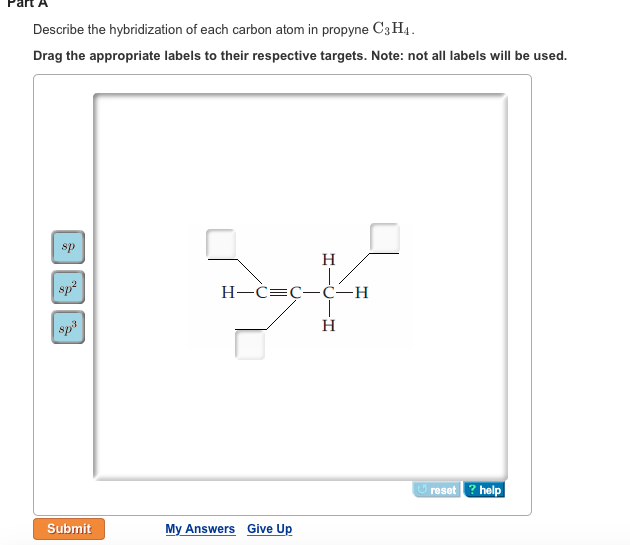
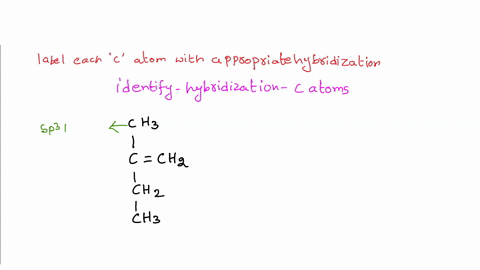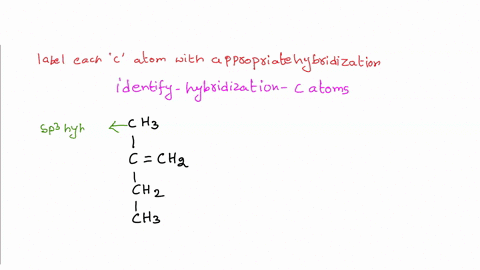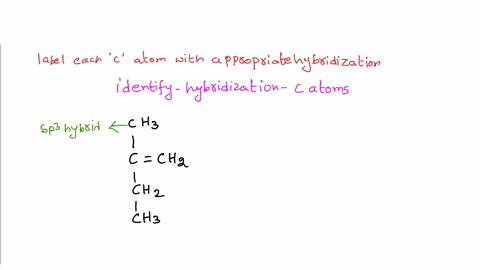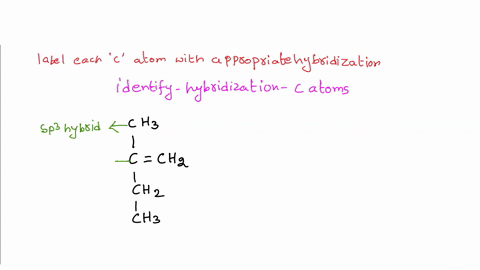
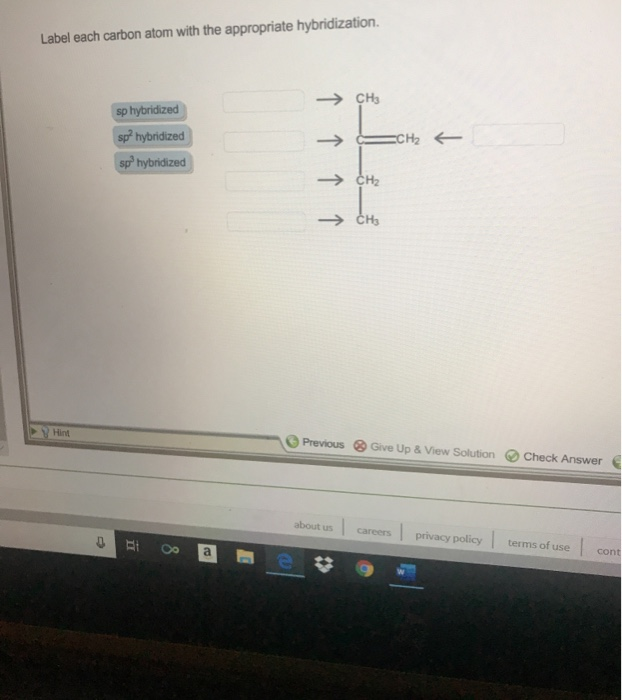
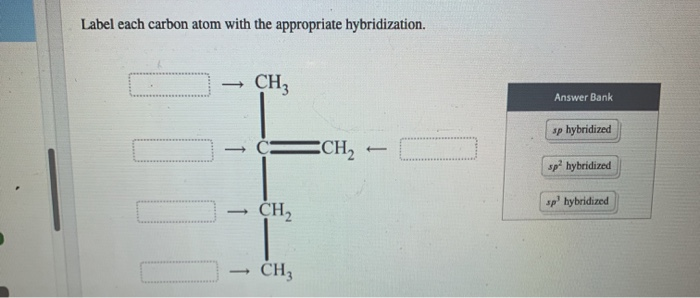
39 label each carbon atom with the appropriate hybridization.
OneClass: Label each carbon atom with the appropriate hybridization.sp Label each carbon atom with the appropriate hybridization.sp,sp2 or sp3 Label each carbon atom with the appropriate hybridization. sp,sp2 or sp3 Show full question + 20 For unlimited access to Homework Help, a Homework+ subscription is required. Verified Answer Tod Thiel Lv2 21 Apr 2020 Unlock all answers Get 1 free homework help answer. Can you label each carbon atom with the appropriate hybridization .... C-C bonded C are sp3 hybridized. In this compound one C=C present and hence both carbons are sp2 hybridized. Left all the C atoms have four single bond around each C atom. so left all the C atoms are sp3 hybridized. number of electrons pairs around sp3 hybridized C atoms = 4 and number of electrons pairs around sp2 hybridized C atoms = 3
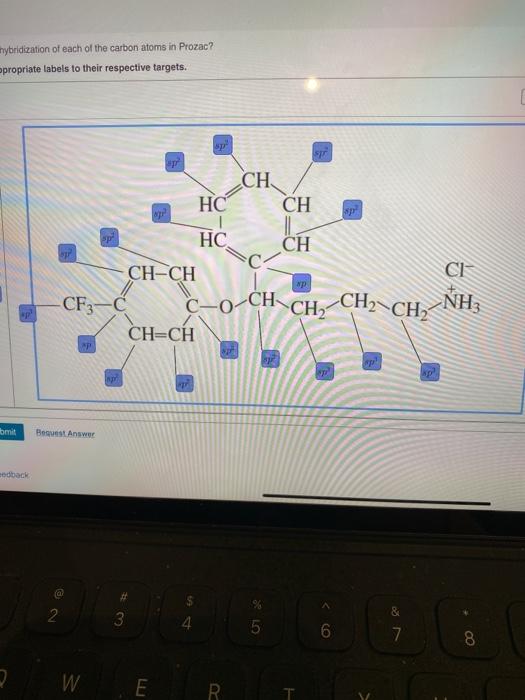
Label Each Carbon Atom With The Appropriate Hybridization. And so, the fast way of identifying a hybridization state, is to say, okay, that carbon has a double bond to it; Label each carbon atom with the appropriate. This is asked in the given right the structure first and then apply the label to each carbon atom.

Label each carbon atom with the appropriate hybridization.
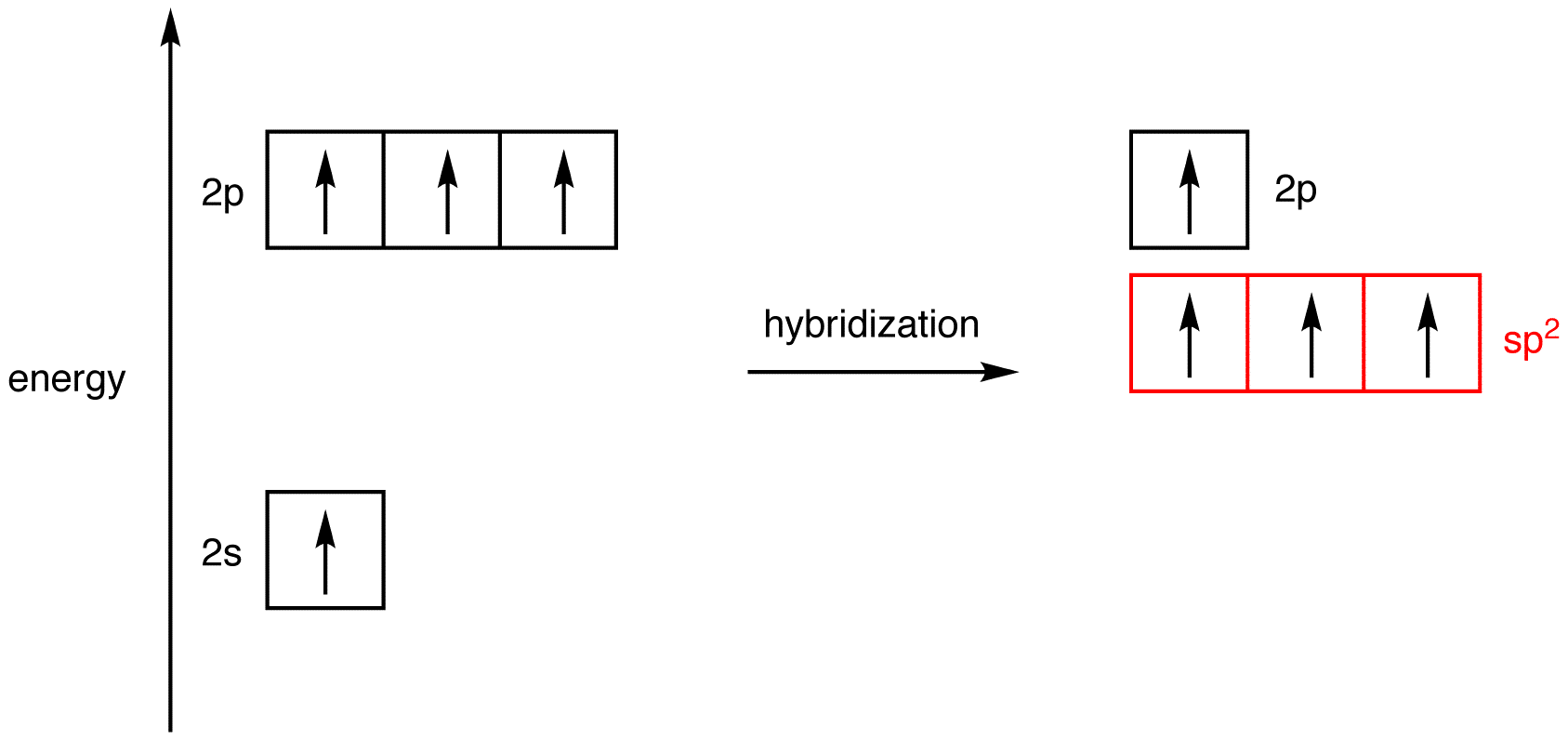
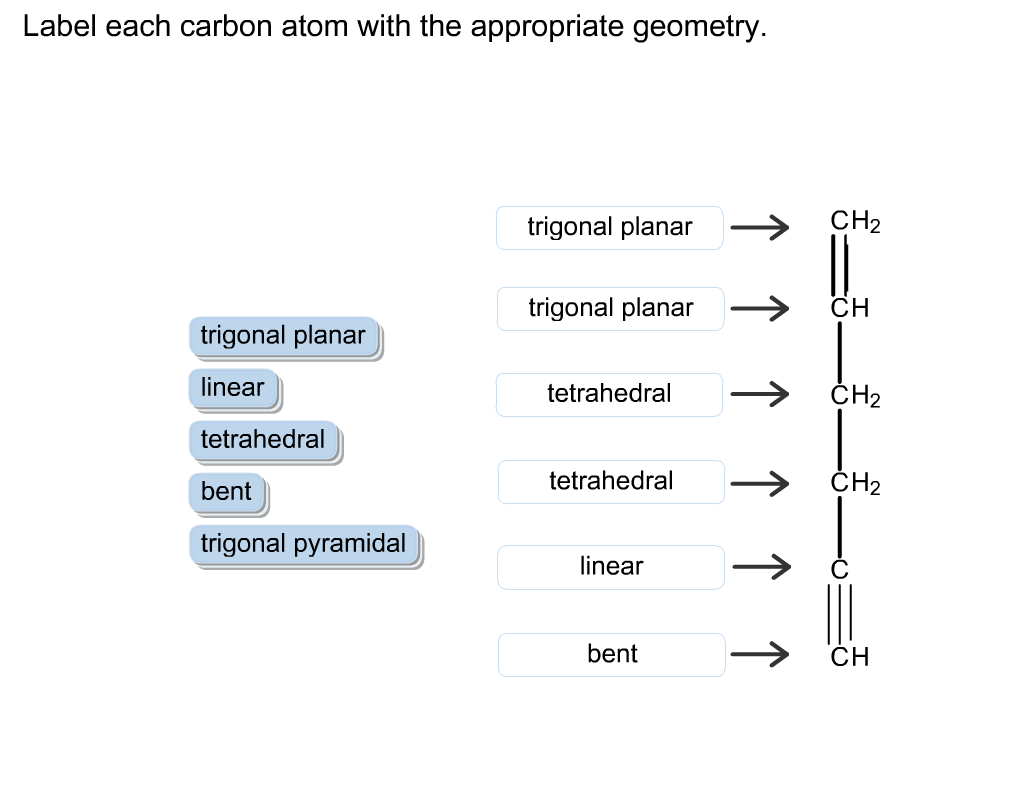
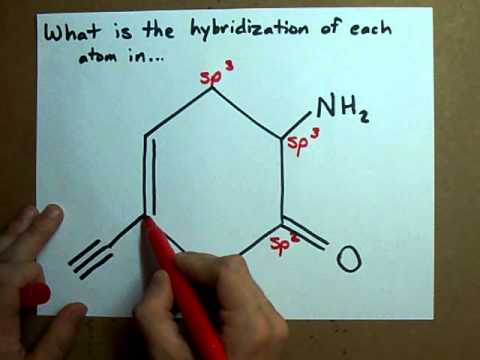
Label each carbon atom with the appropriate geometry. The hybridizations of each carbon in the molecule are as follows If the carbon has 4 single bonds, then the hybridization is. If the carbon has one double bond and two single bonds, then hybridization is. If the carbon has one triple bond or two double bonds, then the hybridization is. What is the hybridization of each atom in this molecule? What is the hybridization of each atom in this molecule? 327,449 views Sep 16, 2011 5.1K Dislike Share Save chemistNATE 223K subscribers More free chemistry help videos:... What is the hybridization of each carbon in this molecule? The 2s and the three 2p orbitals hybridise together and each orbital will be completed by adding one more electron from sharing with N, H, H, and the other C. While the second carbon is sp2 because it forms a double bond with oxygen. In sp2 hybridisation the 2s orbital hybridises with only two 2p orbitals leaving the other 2p orbital unhybridised.
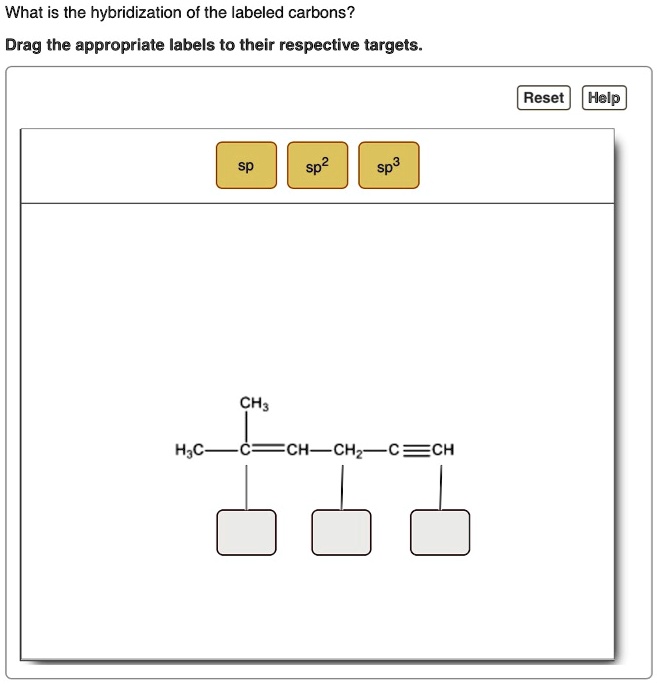
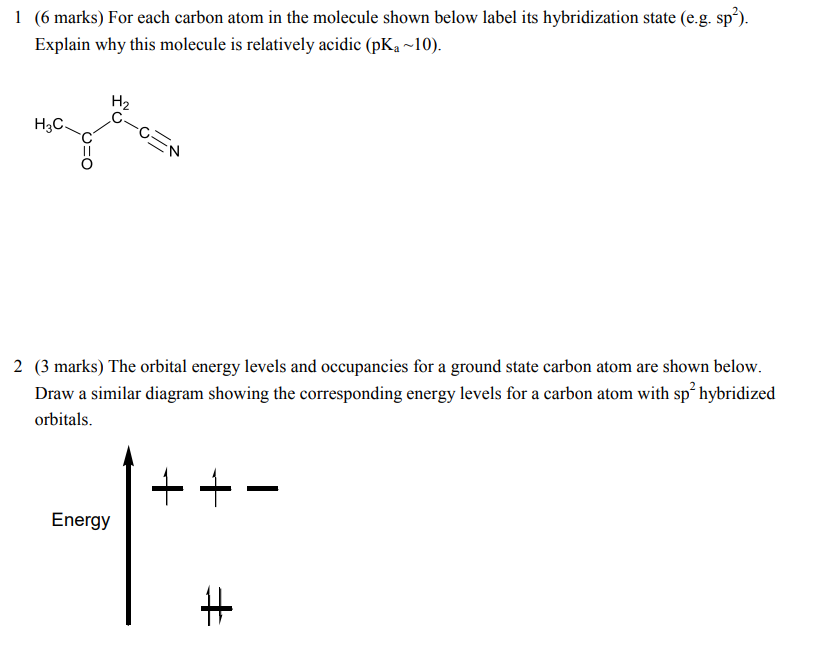
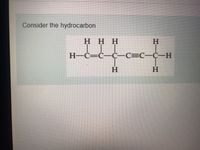
Label each carbon atom with the appropriate hybridization.. Chapter 9 Homework Flashcards | Quizlet There are 6 C atoms in the molecule. Starting on the left, the hybridizations are: sp2, sp2, sp3, sp, sp, sp3. All single bonds are bonds. Double and triple bonds each contain 1 bond. This molecule has 8 C-H bonds and 5 C-C bonds, for a total of 13 bonds. Double bonds have 1 bond and triple bonds have 2 bonds. This molecule has a total of 3 bonds. Hybridization - sp, sp2, sp3, sp3d, sp3d2 Hybridized Orbitals, Examples The shape of the molecule can be predicted if the hybridization of the molecule is known. The bigger lobe of the hybrid orbital always has a positive sign, while the smaller lobe on the opposite side has a negative sign. Try This: Give the hybridization states of each of the carbon atoms in the given molecule. H 2 C = CH - CN HC ≡ C − C ≡ CH label each carbon atom with the appropriate hybridization Label each carbon atom with the appropriate hybridization. sp,sp2 or sp3 raspberrybear867 Label each carbon atom with the appropriate geometry. Label each carbon atom with the appropriate geometry. salmonmink498 4) Draw, and label the hybridization, and bond angles of each carbon atom in: a) toluene b) butane c) acetic acid d) methyl cyanate Hybridization of Carbon - Molecular Geometry and Bond Angles - BYJUS Hybridization of Carbon - Molecular Geometry and Bond Angles Hybridization of Carbon - C atom is either sp, sp2, or sp3 hybridized. Understand the types of hybridization of carbon with molecular geometry and bond angles. Login Study Materials NCERT Solutions NCERT Solutions For Class 12 NCERT Solutions For Class 12 Physics
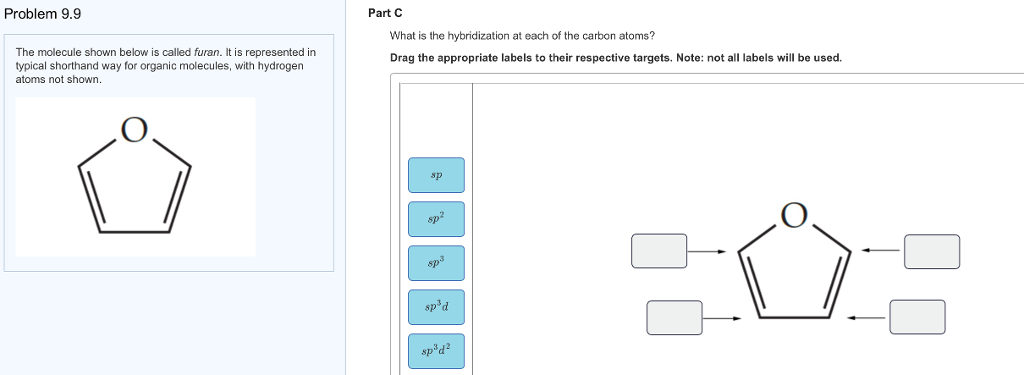
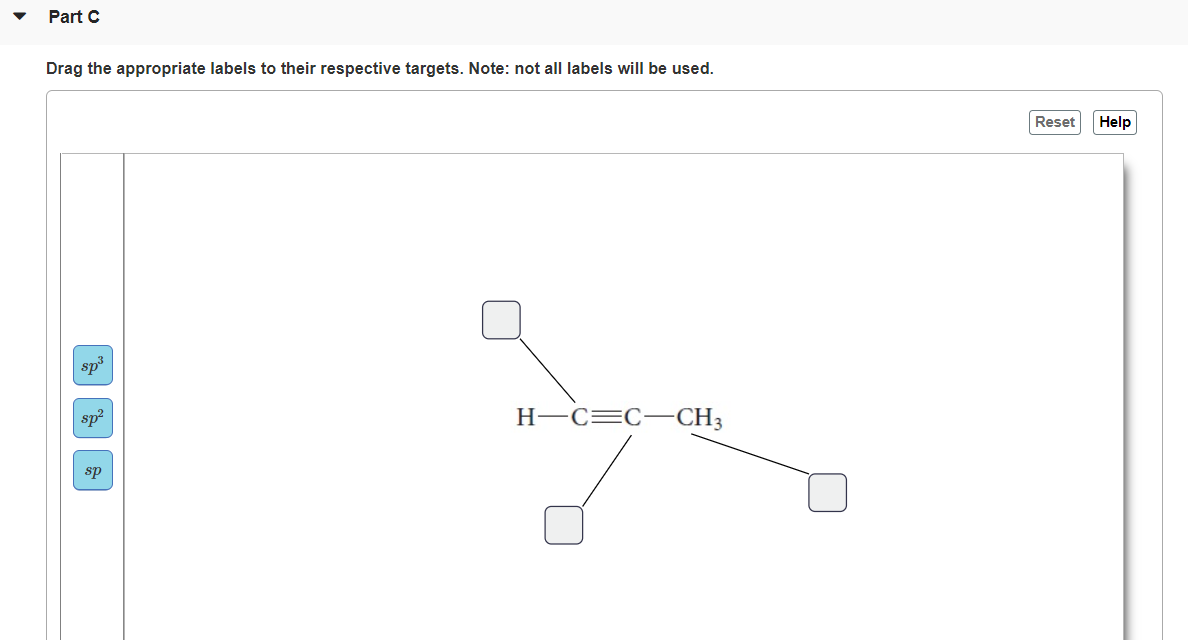
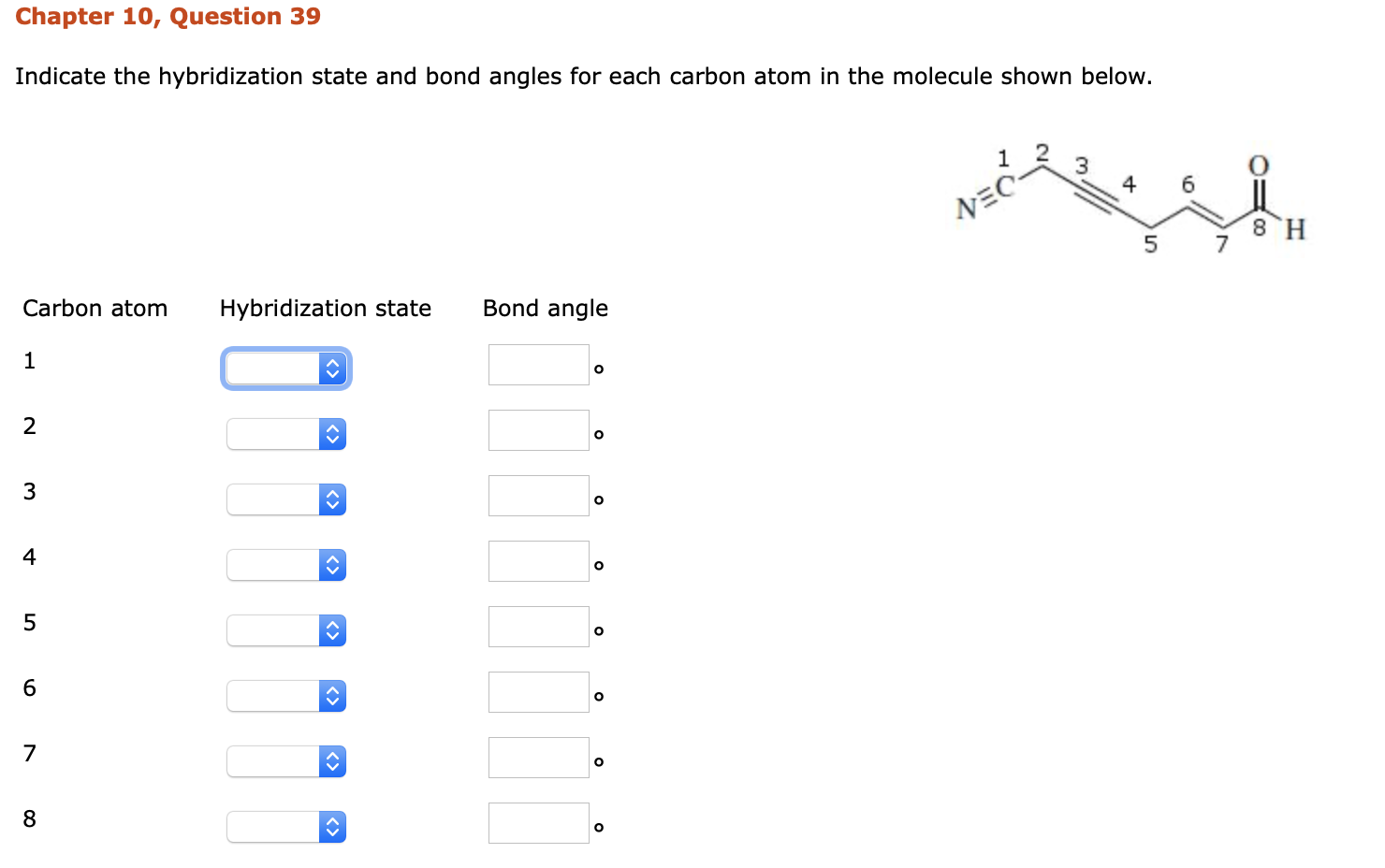
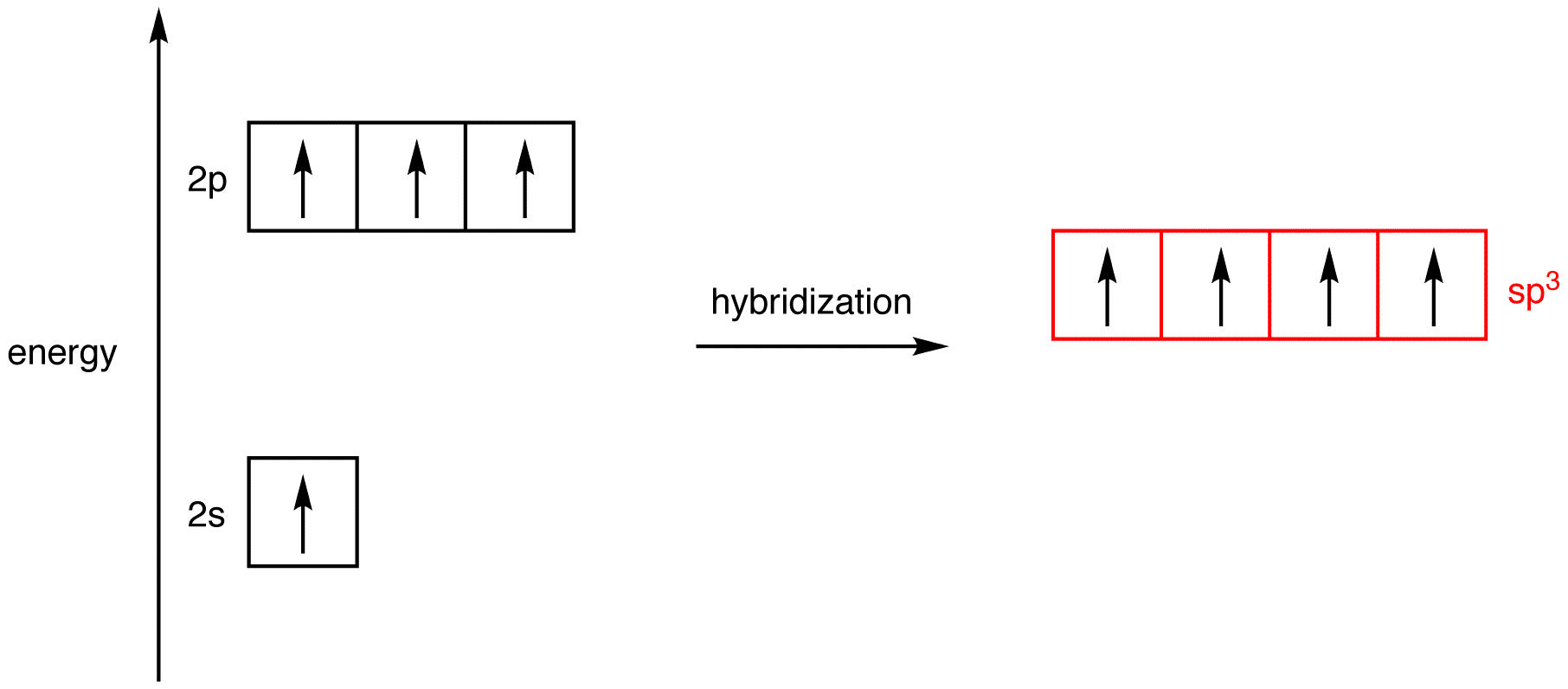
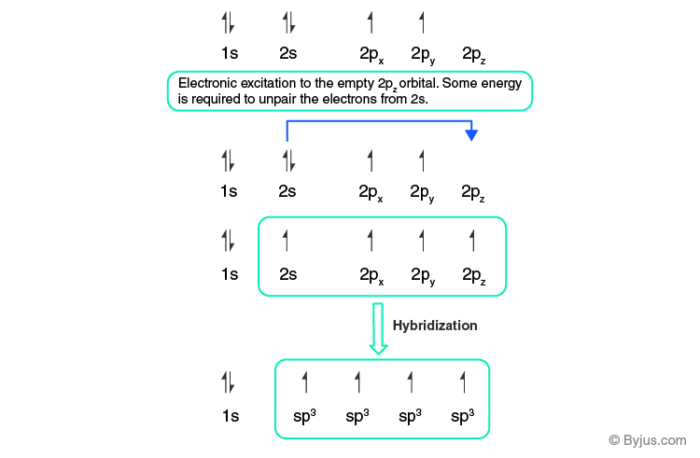
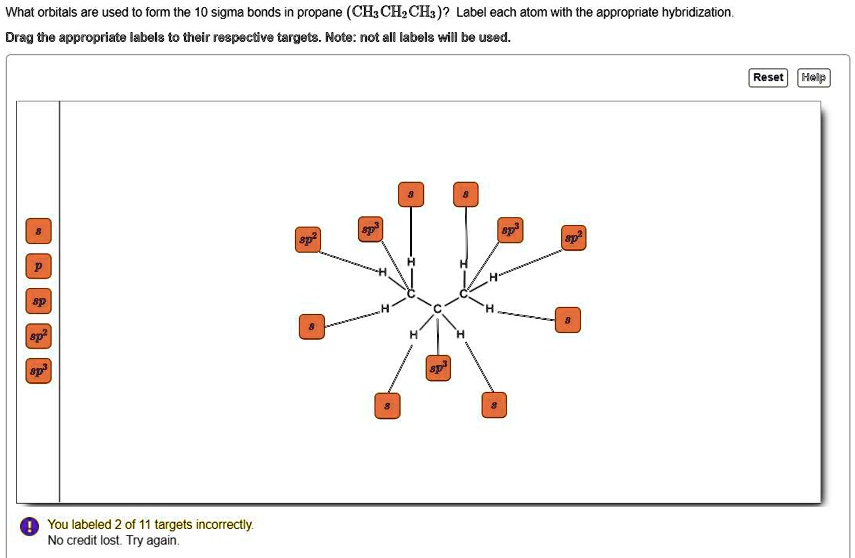
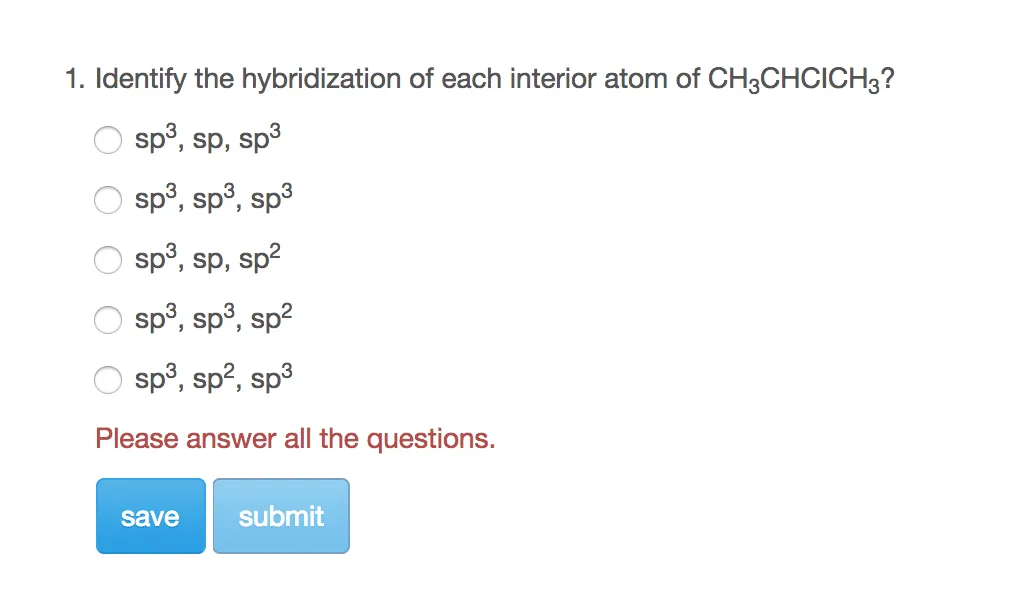
Label the hybridization of each atom noted: hybridization of oxygen ... The hybridization of carbon atom in an alkyne.... what is the hybridization of each carbon atom in acetonitrile... Give the (a) bond order and (b) hybridization state of each carbon atom of carbon monoxide (CO)... bridization, name 3 different compounds that showcase 3 different hybridization variants in the carbon atom. Include ... New questions in Chemistry - Brainly.com Two carbon-carbon single bonds, and eight carbon-hydrogen single bonds. Each of these single bonds is also known as a sigma bond. Every hydrogen atom in this molecule uses an s-orbital to form bonds. Each carbon atom in this molecule forms four sigma bonds. The electrons of each carbon atom are found in one s-orbital and three p-orbitals. OChem Spring 2017 Exam 1 Flashcards | Quizlet Label each carbon atom with the appropriate hybridization (cover bottom) ... Sapling Hw Ch 1.18. a) 120° b) 109.5° Label each carbon atom with its optimum C-C-C bond angle (cover bottom) Sapling Hw Ch 1.19. Rank the following compounds according to increasing positive character of the carbon atom CH₃F What is the hybridization at each of the carbon atoms of the molecule ... The carbon- carbon sigma bond is formed from overlap of an sp3 hybrid orbital on each C atom. ... In sp3 hybridization, the carbon atom is bonded to four other atoms. In this case, 1 s orbital and 3 p orbitals in the same shell of an atom combine to form four new equivalent orbitals. The arrangement of orbitals is tetrahedral with a bond angle ...
label each carbon atom with the appropriate hybridization label each carbon atom with the appropriate hybridization - YouTube 0:00 / 1:54 label each carbon atom with the appropriate hybridization 457 views Jun 11, 2020 5 Dislike Share Save OneClass... Label each carbon atom with the appropriate hybridization This review ... Label each carbon atom with the appropriate hybridization This review was not. Label each carbon atom with the appropriate. School San Diego Mesa College; Course Title CHEM 200; Type. Notes. Uploaded By mertsianadareishvili. Pages 48 This ... What is the hybridization of each carbon in this molecule? The 2s and the three 2p orbitals hybridise together and each orbital will be completed by adding one more electron from sharing with N, H, H, and the other C. While the second carbon is sp2 because it forms a double bond with oxygen. In sp2 hybridisation the 2s orbital hybridises with only two 2p orbitals leaving the other 2p orbital unhybridised. What is the hybridization of each atom in this molecule? What is the hybridization of each atom in this molecule? 327,449 views Sep 16, 2011 5.1K Dislike Share Save chemistNATE 223K subscribers More free chemistry help videos:...
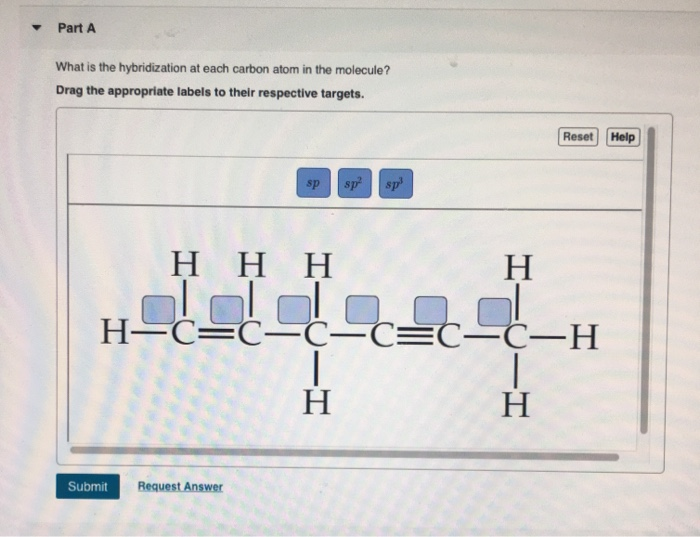
Label each carbon atom with the appropriate geometry. The hybridizations of each carbon in the molecule are as follows If the carbon has 4 single bonds, then the hybridization is. If the carbon has one double bond and two single bonds, then hybridization is. If the carbon has one triple bond or two double bonds, then the hybridization is.


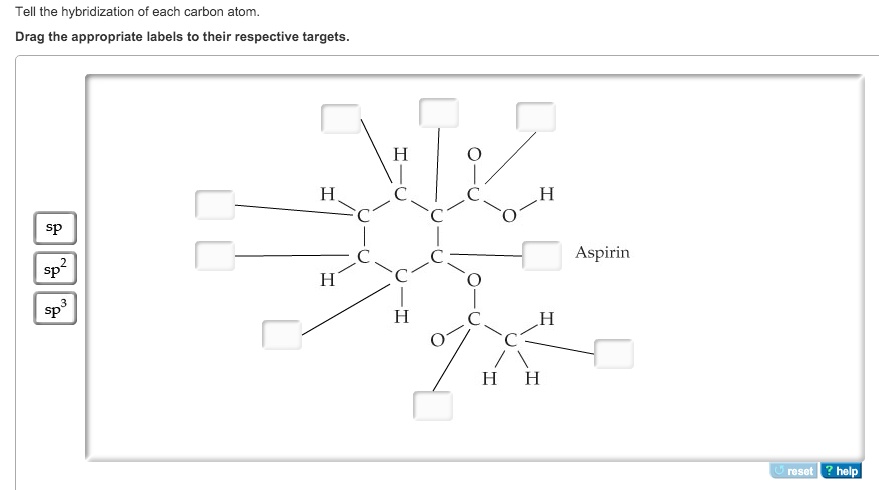

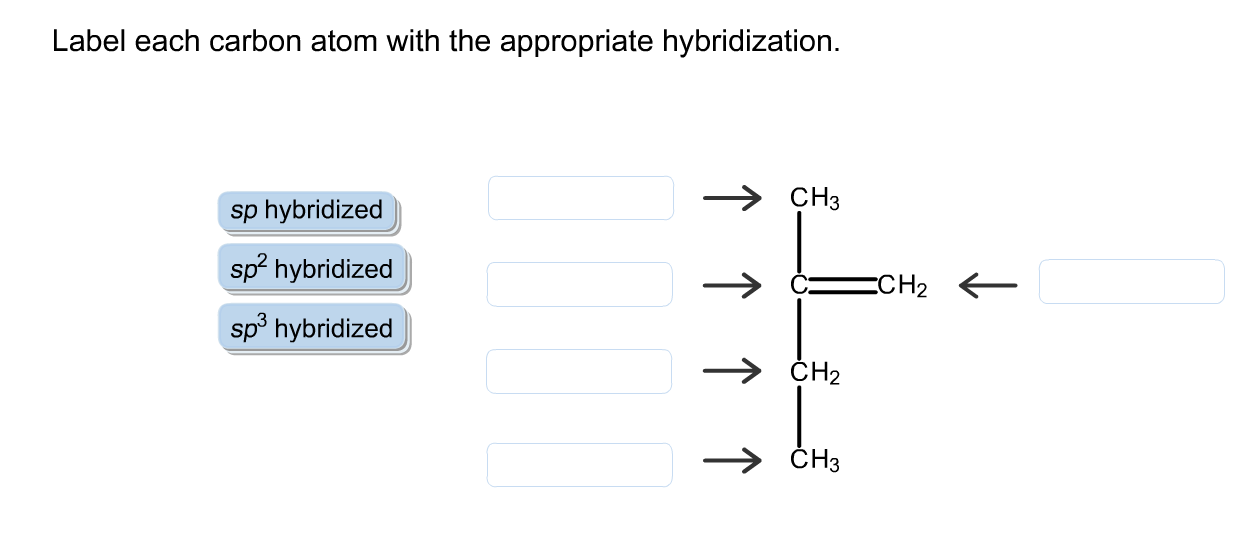
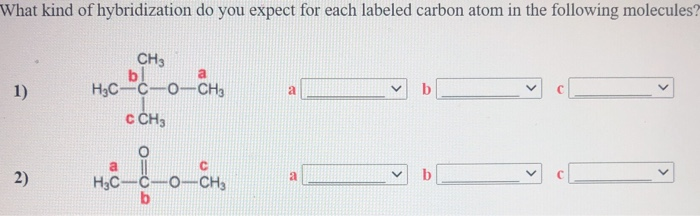
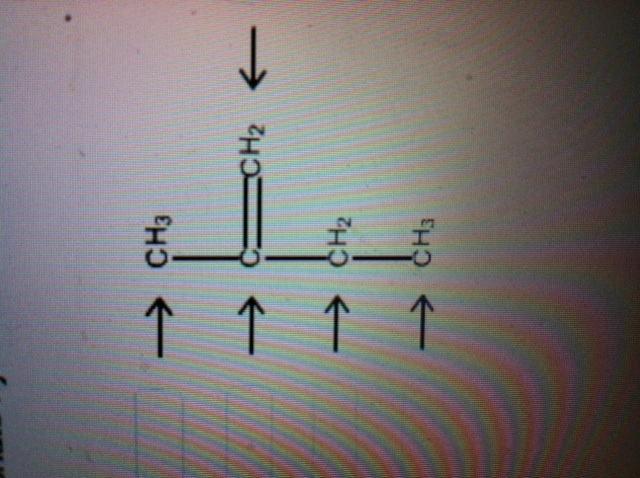
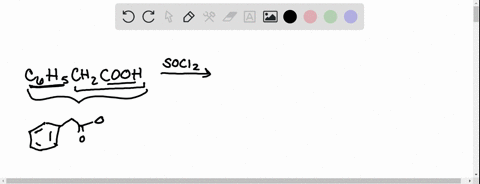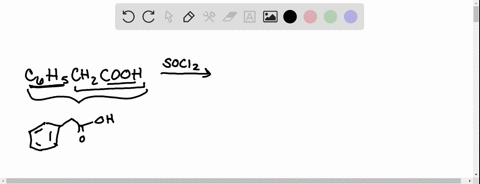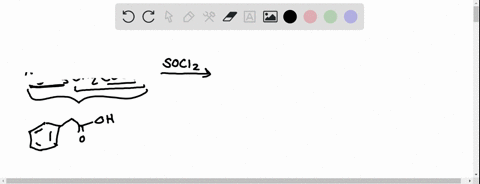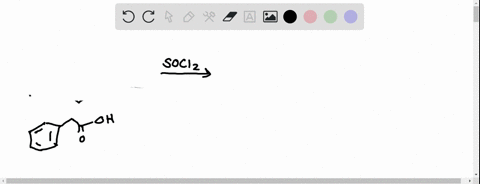
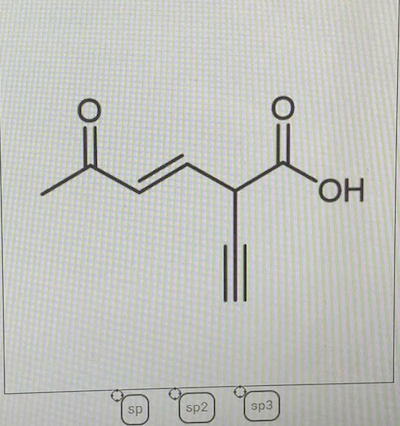
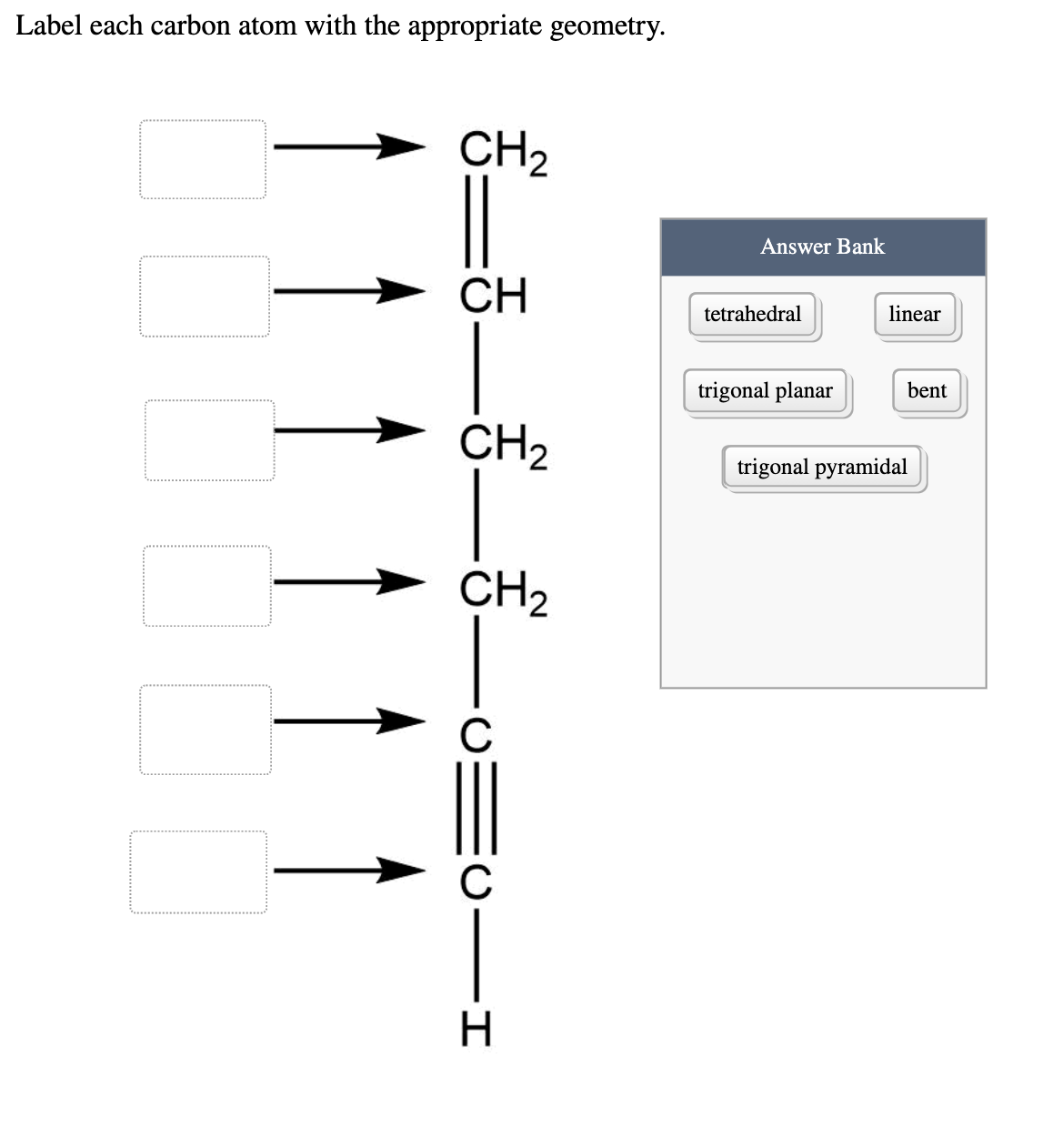
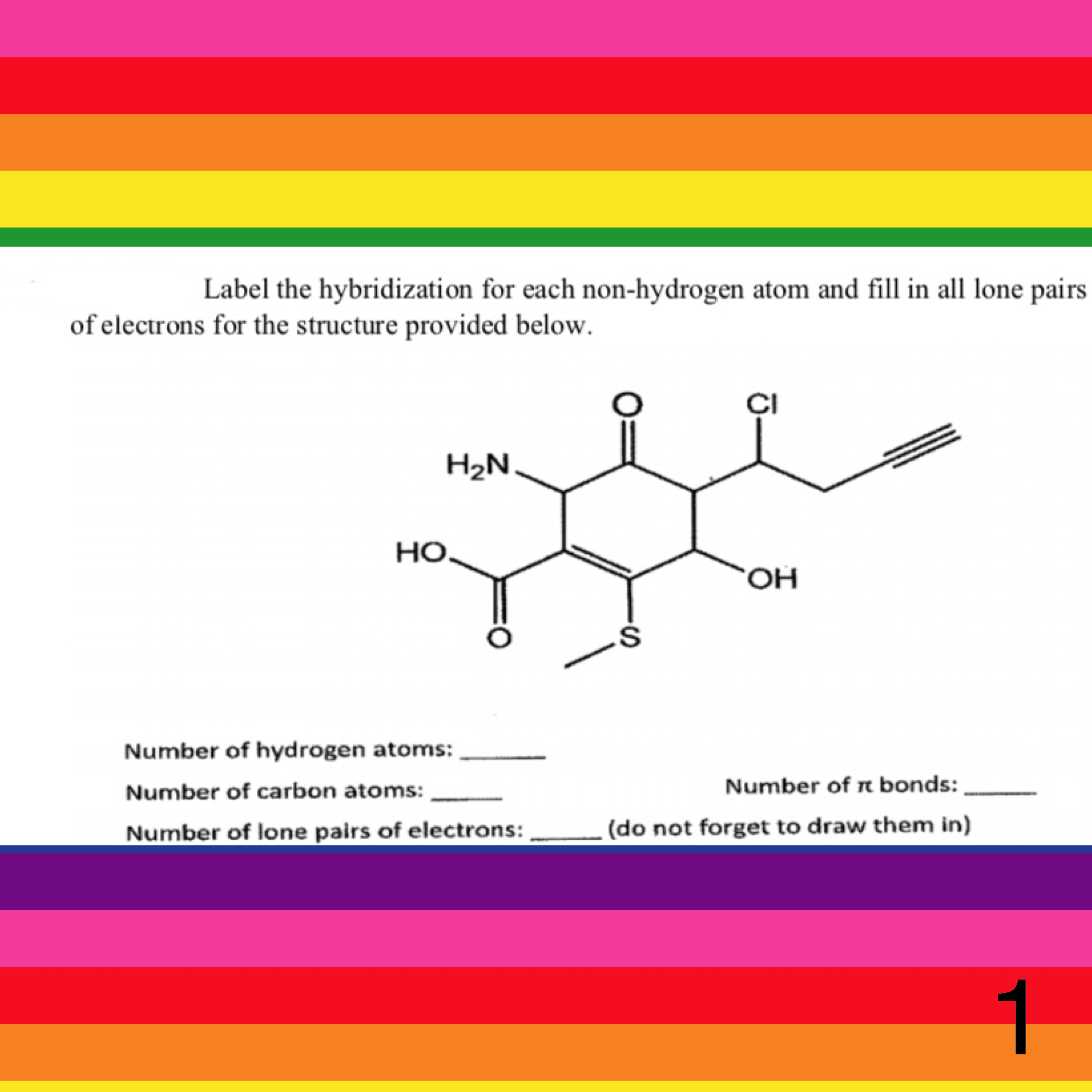


0 Response to "39 label each carbon atom with the appropriate hybridization."
Post a Comment